Jon D. Lurie
INTRODUCTION
Opioids have been used medicinally for thousands of years.1 Throughout that time there has been a constant tension between their ability to relieve pain and suffering and the potential for severe personal and societal harm related primarily to risks of dependency and addiction. As early as the 5th to 3rd centuries BC, medical controversies raged between those extolling the benefits and those warning of the dangers of using opioids.1 A recent dramatic increase in opioid prescriptions and in prescription opioid abuse has reinvigorated this controversy once again. This issue is particularly relevant to the treatment of low back pain given its high prevalence worldwide; in the United States (US), opioids have become the most commonly prescribed class of drugs to treat back pain and over half of all patients on long-term opioids report back pain.2,3 This chapter summarizes the recent epidemiology of opioid prescribing and the evidence regarding effectiveness and potential harms of using opioids for treating back pain.
EPIDEMIOLOGY
Trends in Opioid Prescribing
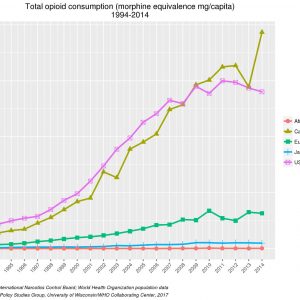
Over the past 30 years, we have seen a dramatic rise in opioid prescribing, particularly in North America, with US prescription opioid sales increasing 4-fold between 1999 and 2010.4 In the US, 245 million prescriptions for opioids were dispensed in 2014 making them the most commonly prescribed class of medications.5 Expenditures on opioids specifically among patients with spine problems increased 660% from $246 million in 1997 to $1.9 billion in 2006.6 In primary care, the major source of opioid prescribing, low back pain is the most common reason for opioid prescription.1 While these increases have been the largest in the US and Canada, substantial increases have also been seen in Europe, with smaller increases in Asia (Fig. 2-1). The rate of increase visibly accelerated after 1996, corresponding to the appearance of new drug formulations and extensive marketing by pharmaceutical companies.4 The majority of opioids in terms of dose equivalents are dispensed to patients on long-term opioids for chronic pain.4
The increase in opioid prescribing was accompanied by a concomitant increase in opioid abuse.1 The US has seen a dramatic rise in admissions for substance abuse treatment and opioid overdose deaths.4 In 2014 alone, 28,000 deaths in the US were attributed to opioid abuse and overdose.7 Almost 40% of drug-overdose deaths in the US in 2013 were related to prescription opioids, double the number attributed to heroin.5 Increases in opioid related deaths have also been seen in the UK and elsewhere.4 Physician prescriptions have become the major source of opioids that are misused for non-medical purposes.5 The increasing awareness of the role that prescription opioids play in the epidemic of opioid addiction, misuse and overdose deaths have led to a careful re-examination of the appropriate use of opioids, particularly in relation to the treatment of chronic pain.7
Geographic Variation in Use of Opioids
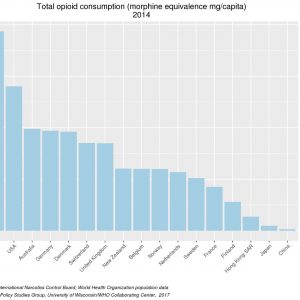
Large amounts of geographic variation in the use of health services often relates to a lack of consensus by physicians about which services are appropriate and/or necessary.8 Studies that have reported on the proportion of patients with chronic low back pain treated with opioids show wide variation, ranging from 3% to 66% depending on the setting.9 Figure 2-2 shows the dramatic variation in prescription opioid use among selected countries in 2014. While per capita use of these strong opioids is highest in North America and much lower in Asia, there is substantial variability among European countries. Similarly, within the US, there is 23-fold variation in per capita opioid prescriptions within Medicaid programs between states.4 These differences highlight the lack of strong evidence regarding the efficacy, safety and optimal prescribing practices for opioids.4
Adverse Selection in Opioid Prescribing
The lack of strong medical consensus on the proper indications for long-term opioid use leads to non-medical factors driving the decision of who does and does not receive opioid analgesia. In studies looking at the characteristics of patients with back problems receiving opioids compared to those not receiving opioids, those with worse pain and function were more likely to receive opioids as would be expected; however, many other characteristics were also associate with receipt of an opioid prescription including higher expressed distress and suffering,9 a history of substance abuse,10 as well as obesity, tobacco use and worse mental health.11 This results in a phenomenon known as “adverse selection” as patients with history of substance-use disorder and negative affect (depression and anxiety) have less pain improvement with opioids12,13 and greater risk for adverse events of overdose and addiction.5
EFFICACY AND EFFECTIVENESS OF OPIOIDS FOR BACK PAIN
Acute Low Back Pain
Opioids are typically the strongest and the most effective analgesics available for most causes of acute pain.7 Nevertheless, there is very little data on their effectiveness for back pain specifically. A recent systematic review of pharmacologic treatments for low back pain identified only one randomized trial of opioids in acute low back pain, which found no difference in pain or function between oxycodone/acetaminophen compared to placebo in patients also given naproxen (both groups).14 A number of observational studies in injured workers with acute low back pain found that those receiving early treatment with opioids generally had longer work disability.4 Whether this is due to confounding by indication – patients with worse injuries being more likely to receive opioids – or the negative effects of opioids, perhaps through their “deactivating” effects limiting physical activity, is unclear.15 Similar results were found in primary care patients with acute low back pain in the UK with those receiving opioids showing worse functioning at 6 months.4 Thus, while opioids are clearly strong analgesics and while there is very little data specifically evaluating them in acute low back pain, there is no evidence that they improve the course of acute low back pain and if used should probably be limited to a few days-worth of treatment as is generally indicated for routine trauma and other acute injuries.15
Chronic Low Back Pain
There are many more trials evaluating the effectiveness of opioids in chronic low back, than for acute low back pain, however the evidence remains of relatively low quality; ironically most of the studies evaluated only short term outcomes and many had other significant limitations such as high drop-out rates.4 A number of systematic reviews on the topic over the last 10 years have consistently concluded that the evidence is of low to moderate quality showing that opioids provide better pain control in the short term compared to placebo but the effects on function and on long term outcomes are unclear.4,9,14,16,17
The most recent Cochrane Collaboration review included 15 trials comparing opioids to placebo for chronic low back pain.16 There were 6 trials of strong opioids (tapentadol, morphine, oxymorphone, hydromorphone and oxycodone) included in the meta-analysis of almost 1900 subjects; approximate mean doses ranged from 60 to 250 morphine equivalents per day and the follow-up from 12-15 weeks. Overall, they found that strong opioids had moderate short-term efficacy for reduced pain and small short-term efficacy for improved function compared to placebo. Two trials with 272 subjects compared strong opioids to antidepressants; meta-analysis found very-low quality evidence of no difference in pain or functional outcomes. Two other trials with a total of 653 subjects evaluated transdermal buprenorphine, a partial opioid agonist/antagonist, compared to placebo. There was very-low quality evidence that transdermal buprenorphine at doses of 20 mcg/hr up to a maximum of 40 mcg/hr was better than placebo in reducing pain with no difference in functional outcomes.
In the same review, Tramadol plus or minus acetaminophen was evaluated in 5 trials with almost 1400 subjects.16 Meta-analysis found low-moderate quality evidence that tramadol improved pain and function in the short term, with standardized response means of similar magnitude to those found for strong opioids. They also found 2 trials comparing tramadol to celecoxib at 6 weeks. There was very-low quality evidence that tramadol was better at reducing pain than celecoxib, although the number of subjects completing the trial was significantly higher in the celecoxib group (86% versus 72%) and no functional outcomes were studied. With similar evidence of efficacy and an apparent lower risk of addiction than strong opioids, some have recommended tramadol as a potentially safer option when opioid therapy is being considered in chronic low back pain.15 Caution is warranted, however, given that a recent epidemiologic study found the likelihood of continued opioid use at 1 and 3 years was higher when an initial opioid prescription was for tramadol (13.7 and 6.8%, respectively) compared to short-acting hydrocodone (5.1 and 2.4%, respectively).18 This may indicate a lower threshold for continuing this medication due to lower perceived risk, a selection of tramadol in patients who are expected to need to continue for long term use due to perceived lower risk, or it may indicate that the risk is not a low as thought.
In summary, opioids appear to provide modest short-term pain relief and less reported difficulty with daily activities in the short-term relative to placebo in patients with chronic low back pain; however, there is almost no data on based on objective physical function measures and no randomized trial data beyond 4 months.16,17 Observational studies on the efficacy of long-term opioids find that among long-term users only about a quarter maintain significant reductions in pain and 16% improved function, and long-term opioid users generally report worse pain and quality of life than chronic pain patients who are not taking opioids.4 These data must be interpreted cautiously as they may be confounded by the indications prompting the use of long-term opioids. Nonetheless, there is little to no evidence that long-term opioids are more effective than other analgesics for chronic low back pain and any potential benefits need to be weighed against the potential for serious adverse effects including misuse, abuse, addiction, overdose and death.16
ADVERSE EFFECTS OF OPIOID TREATMENT
Adverse Effects
Opioids have a number of side effects that make them difficult to take for many patients. In fact, many controlled trials of opioids use “enriched enrollment” designs to try to address high drop-out rates related to adverse effects and lack of efficacy.4 Common adverse effects are shown in Table 2-1 and generally can be grouped in gastrointestinal, cardiovascular, endocrine and central nervous system effects. Across the set of adverse events, the number needed to harm in short-term trials for any adverse event was only 4.2 – for approximately every four patients treated, one would have an adverse event.4
| Symptomatic Side Effects | Longer-Term Risks/Harms |
|---|---|
|
|
Constipation is the most common side effect and while some initial adverse effects improve with time, constipation often does not and frequently requires long-term management for patients on chronic opioids.4 Other gastrointestinal effects include nausea, vomiting, depressed appetite and dry mouth.
Opioids have also been associated with adverse cardiovascular outcomes. An evidence synthesis as part of the CDC Guideline for Prescribing Opioids for Chronic Pain found increased risk of myocardial infarction (Odds ratio [OR] 1.28) in current opioid users versus non-users, and a stepwise increase in risk associated with increasing dose.19
Chronic opioid therapy can have a variety of adverse endocrinologic effects including androgen deficiency, erectile dysfunction, oligo- or amenorrhea, infertility and osteoporosis.
Opioids effect the hypothalamus, reducing levels of gonatropin releasing testerosterone and estradiol levels, both hormone reducing, leading to potential adverse effects in both men and women.4 Epidemiologic studies have shown that up to 20% of men on chronic opioids received testosterone replacement or medication for erectile dysfunction with an odds ratio > 1.5 compared to similar men no taking opioids.20 Menstrual irregularities and probable reduced fertility have been observed in association with 48% to 57% reductions in testosterone, estradiol and dehydroepiandrosterone sulfate values in opioid-consuming women compared to controls.21 High rates of osteoporosis have also been observed in patients on chronic methadone.1 Androgen deficiency may be greater with long-acting/extended release opioids as compared with short-acting/immediate release formulations.19
Central nervous system adverse effects are also common problems in patients taking opioids. Sedation and dizziness occur in 20-30% and combined with the endocrinological effects on bones may contribute to dose related effects on falls and fractures.4 There are also cognitive effects on attention and reaction time. This may increase in the risk of motor vehicle trauma; however, the data are unclear.4,19 Opioid use is associated with depression, with increased incidence being associated with both dose and duration; however, the direction of causation is unclear as depression may lead to increased use of opioids as well as vice-versa.4 Opioid use is also associated with sleep disturbances particularly sleep disordered breathing.22
The other major central nervous system effects of opioids include tolerance and dependence. Tolerance and physical dependence and expected and nearly inevitable consequences of prolonged administration of opioids.5,15 Tolerance denotes decreased efficacy of a drug over time leading to, in this case, a decrease in opioid potency with repeated use.5,23 Notably, tolerance to the analgesic and euphoric effects of opioids can develop quickly while tolerance to the respiratory depressive effects develop more slowly, which contributes to the risk of overdose deaths with opioids.5 Physical dependence denotes the appearance of symptoms with the abrupt discontinuation of opioids; typical withdrawal symptoms include pain (diffuse muscle aches and abdominal pain), nausea, vomiting, piloerection, tachycardia, agitation, diarrhea, chills and insomnia.5,15 These symptoms can vary substantially in severity and duration depending on the individual and the context (type, dose and duration) of opioid use but generally resolve within 1-2 weeks of discontinuation and typically require tapering rather than abruptly discontinuing opioids after prolonged use.5,15 Psychological dependence includes symptoms of anhedonia, dysphoria and opioid cravings, which can persist long beyond the resolution of physical dependence.15 The timeline for the development of dependence can differ substantially between individuals, but data suggest that clinically withdrawal symptoms can be induced with even a single parenteral dose opioid and clinically important dependence in just one month of daily opioid use.4
A possibly related but distinct phenomenon is opioid induced hyperalgesia. Opioid induced hyperagesia is heightened pain sensitivity caused by opioid use leading to the paradoxical response of higher doses of opioids leading to more pain.5,23 The true incidence and the exact underlying pathophysiology of opioid induced hyperalgesia remain unclear, but the phenomenon has been demonstrated in both animal and human studies.23 Opioid induced hyperalgesia may be confused with tolerance where increasing the dose may be appropriate; however, dose escalation leads to worsening of symptoms in hyperalgesia.23 The treatment for opioid induced hyperalgesia is dose tapering or tapering and discontinuation of opioids.5
Opioid Use Disorder
As opposed to the develop of tolerance and physical dependence, which are predictable, iatrogenic consequences of regular opioid use in nearly all patients, opioid use disorder and addiction occur in only a subset of people exposed to opioids, but when they do develop they represent a distinct chronic medical problem even after opioid use is discontinued.4,5 The physiologic basis of addiction appears to be distinct from those of tolerance and physical dependence, and it is characterized by features including: drug cravings, obsessive thinking about the drug, loss of inhibitory control over efforts to avoid drug use, and compulsive drug taking despite negative consequences.5 The persistence of opioid use disorder long after discontinuation of opioids is clinically important because of the associated high risk of overdose after a period of abstinence when tolerance resolved, resulting in very high risks of overdose among patients with opioid addiction upon release from prison or drug detoxification programs.5
Because of confusion regarding the use of terms representing the spectrum of problems related to opioid use including tolerance, dependence, diversion and addiction, the prevalence of opioid use disorder can be difficult to reliably assess with different studies using widely different definitions.4 Rates of strictly defined addiction appear to be in the 1-8% range, while rates of problematic use including a variety of behaviors including misuse, abuse and other problematic behaviors in the 24-31% range.4,5 Previous misconceptions that pain somehow protected people from opioid addiction and that the use of opioids to treat chronic pain could occur with vanishing low rates of addiction have been thoroughly debunked.
Table 2-2 shows known risk factors associated with higher risk for addiction. Much of the risk appears to be genetically modulated with estimates of heritability similar to hypertension and diabetes.5 Adolescents appear to be at increased risk, perhaps because of increased brain plasticity and/or underdeveloped executive brain functions, and particular care should be taken when making prescribing decisions in this group.5 Not surprisingly, increased dose and duration of exposure also increases the risk in all groups. As discussed above, even though prior substance use disorder and other comorbid mental health disorders are known to increase the risk of addiction, they are also associated with increased likelihood of receiving an opioid prescription for low back pain. This phenomenon of so-called “adverse selection” likely also leads to a substantial overestimation of benefit and an underestimation of harm in clinical trials of opioid treatment, where a history of substance abuse or comorbid mental illness are typically exclusionary, compared to actual clinical practice.4
| Patient Factors |
|
| Treatment Factors |
|
Overdose and Death
Opioid overdose and related deaths have increased dramatically worldwide over the past few decades. From 2000–2014 in the US, the age-adjusted rate of drug overdose death involving opioids nearly tripled, from around 3 per 100,000 persons in 2000 to 9 per 100,000 in 2014, leading to over 28,600 opioid overdose deaths in 2014 alone.24 Recently, unintentional overdose deaths among adults 24–65 surpassed deaths from motor vehicle accidents in both the US and Australia.25 The rate of non-fatal opioid overdoses requiring medical treatment has increased even more dramatically creating a major public health crisis.5
Respiratory depression is the main presentation of opioid overdose and the usual cause of death in fatal events.4 Table 2-3 summarizes the major risk factors associated with increased likelihood of overdose and can generally be divided in to patient factors and treatment factors. The important role of mental health issues, particularly depression and pre-existing substance use disorder, along with increased opioid dose and duration have already been discussed in terms of their potent effects on the risk of addiction and these concerns carry over into the increased risk of overdose and overdose death. Compared to patients prescribed 20 mg per day morphine equivalents or less, those prescribed more than 100 mg per day had a 3 to 9 fold increased risk of serious or fatal overdose.4 In addition to dose and duration, the use of long acting opioid formulations, the combined prescription of opioids and benzodiazepines, use of opioids in patients with sleep-disordered breathing, the use of opioids in patients with renal or hepatic dysfunction, older age, pregnancy and mental health comorbidities increases the overdose risk.19
TABLE 2-3. Factors associated with increased risk of overdose.
| Patient Factors |
|
| Treatment Factors |
|
INFLUENCE OF PRESCRIBING BEHAVIOR ON RISK OF LONGTERM USE
Opioids are most commonly used for short-term treatment of acute pain; however, the largest volume of opioids are prescribed to those receiving long-term opioid treatment of chronic pain.26 Recently a number of studies have begun to look at the characteristics of initial opioid prescribing and their relationship to long-term use. Across a number of studies with somewhat differing methodologies, the risk of long-term opioid use after an initial opioid prescription ranged from 1% to 6%.18,26,27 The main predictors of long-term opioid use after initial prescription was the dose, duration, and use of a long-acting formulation in the original prescription. In a study by Deyo et al., among those who received four or more fills (three or more refills) of their original opioid prescription 26.1% became long-term users compared to 2.9% among those with only a single fill in the initial month; similarly among those with the highest cumulative dose in the initial month (3200 – 3999 MMEs) 46.2% became long-term users compared to 2% for those with less than 120 MMEs during the initial month.26 Overall, those initially prescribed a long-acting opioid had a 24.5% chance of becoming a long-term user compared to 3.5% initially prescribed a short-term opioid. Similarly in a study by Shah et al., the largest increases in probability of long-term use were observed when the initial prescription supply exceeded 10 days, when the patient received a third prescription, or when the cumulative dose exceeded 700 MMEs.18 This same study also showed that continued use at one year was 27.3% among those initially prescribed a long-acting opioid compared to 5% among those initially prescribed short-acting hydrocodone or oxycodone.
In a study of emergency physician practices, there were large variations in the tendency to prescribe opioids; within the same hospital the rate of opioid prescribing between low-intensity and high-intensity prescribers varied, on average, threefold from 7% to 24% of visits.27 The risk of long-term opioid use was significantly higher (OR 1.3) among patients treated by high-intensity providers compared to low-intensity providers as was the rate of subsequent encounters for falls and fractures and for overdose.27 While these data are observational, the use of an instrumental variable based on overall prescribing habits of different physicians within the same hospital minimizes the risk of selection bias confounding these results.
RISK REDUCTION STRATEGIES
The most effective strategy for reducing the risk associated with opioid use in the treatment of back pain is to avoid there use all together given the very limited evidence of effectiveness and the considerable evidence of potential harm. However, there will be occasions for their appropriate use. When these occur, strategies include using the lowest effective dose for the shortest period of time, usually with short-acting opioid formulations, and typically less than 7 days for acute events. Short-term opioid therapy should not become long-term use of opioids without careful consideration.15 When opioids are used for longer-term therapy, careful management involving screening for substance use disorder, using prescription drug monitoring programs, urine drug screening, and clearly delineated doctor-patient agreements on adherence and acceptable medication-related behaviors is recommended.5 The use of a long-acting opioid and additional short-acting opioid for “breakthrough” pain should particularly be avoided when using opioids in the long-term to treat chronic pain.15
CONCLUSION
The use of opioids has increased dramatically worldwide both in general and for back pain in particular. The evidence for the effectiveness of opioids for back pain is limited to only modest effects and only in the short-term. The risks of opioids, however, are extensive and are increased with higher doses, longer duration of use, the use of long-acting formulations such as methadone or extended release formulations of short-acting opioids. The presence of mental health comorbidities or prior substance use disorder decreases the likelihood of opioid effectiveness and increases the likelihood of adverse events. The decision to use opioids for the treatment of back pain must thus be considered very carefully if at all.
The CDC guidelines for prescribing opioids for chronic pain conclude the following:
- Non-pharmacologic therapy and non-opioid pharmacologic therapy are preferred for treating chronic pain;
- Before starting opioid therapy, explicit treatment goals and consideration of how opioid therapy will be discontinued if these goals are not met should be discussed with the patient;
- Discussion of risks and benefits should occur before starting opioids and periodically be revisited during chronic opioid therapy;
- When starting opioids for chronic pain, short-acting formulations should generally be used and the combination of long-acting and short-acting opioids together should be avoided;
- The lowest effective dose should be used, care should be taken when considering a dose of 50 MME per day or more and doses of 90 MME per day or more should generally be avoided;
- Short-term use of opioids in the acute setting should limit the prescription to the lowest effective dose of a short-acting opioid formulation and should prescribe no greater duration than needed, usually less than 7 days and often 3 days or less;
- The effectiveness of opioid treatment should be regularly monitored and tapered or discontinued if appropriate;
- Risks for opioid related harms should be assessed prior to initiation and periodically throughout the course of treatment along with strategies to mitigate risks when present;
- Medication related behaviors should be assessed prior to initiation and periodically throughout the course of treatment using data such as prescription drug monitoring programs where available;
- Urine drug testing should be used prior to initiation and periodically throughout the course of treatment to assess adherence and for substance use disorders;
- Avoid the combination of opioid and benzodiazepines when possible; and
- Evidence-based treatments for opioid use disorder should be arranged for patients when it is identified.19
PEARLS AND PITFALLS
- A dramatic rise in opioid prescribing has been seen over the last 30 years with a concomitant increase in opioid abuse, opioid overdoses, and opioid-related deaths.
- Opioids have a number of side effects and a high rate of medication related adverse events.
- The lack of consensus on the proper indications for long-term opioid use and the marked variability in prescribing habits between providers leads to adverse selection in which patients with history of substance-use disorder and negative affect (depression and anxiety) who have less pain improvement with opioids and greater risk for adverse events of overdose and addiction are more likely to be prescribed opioids.
- Opioids appear to provide modest short-term pain relief and less reported difficulty with daily activities in the short-term relative to placebo in patients with chronic low back pain; however, there is almost no data based on objective physical function measures and no randomized trial data beyond 4 months
- There is little to no evidence that long-term opioids are more effective than other analgesics for chronic low back pain and any potential benefits needs to be weighed against the potential for serious adverse effects including misuse, abuse, addiction, overdose and death.
- The nature of initial opioid prescribing can affect the risk of long-term opioid use; prescriptions should be for the lowest effective dose of a short-acting opioid formulation and should be for no greater duration than needed, usually less than 7 days and often 3 days or less.
- Combinations of opioids and benzodiazepines should generally be avoided as should the concomitant use of short-acting and long-acting opioids.
SUGGESTED READING
- Ballantyne JC. Avoiding opioid analgesics for treatment of chronic low back pain. JAMA. 2016;315(22):2459-2460.
- Deyo RA, Von Korff M, Duhrkoop D. Opioids for low back pain. BMJ. 2015;350:g6380.
- Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645.
- Martell BA, O’Connor PG, Kerns RD, et al. Systematic review: opioid treatment for chronic back pain: prevalence, efficacy, and association with addiction. Ann Intern Med. 2007;146(2):116-127.
- Volkow ND, McLellan AT. Opioid abuse in chronic pain--misconceptions and mitigation strategies. N Engl J Med. 2016;374(13):1253-1263.
REFERENCES
- Katz N. Opioids: after thousands of years, still getting to know you. Clin J Pain. 2007;23(4):303-306.
- Hudson TJ, Edlund MJ, Steffick DE, Tripathi SP, Sullivan MD. Epidemiology of regular prescribed opioid use: results from a national, population-based survey. J Pain Symptom Manage. 2008;36(3):280-288.
- Ivanova JI, Birnbaum HG, Schiller M, Kantor E, Johnstone BM, Swindle RW. Real-world practice patterns, health-care utilization, and costs in patients with low back pain: the long road to guideline-concordant care. Spine J. 2011;11(7):622-632.
- Deyo RA, Von Korff M, Duhrkoop D. Opioids for low back pain. BMJ. 2015;350:g6380.
- Volkow ND, McLellan AT. Opioid abuse in chronic pain--misconceptions and mitigation strategies. N Engl J Med. 2016;374(13):1253-1263.
- Martin BI, Turner JA, Mirza SK, Lee MJ, Comstock BA, Deyo RA. Trends in health care expenditures, utilization, and health status among US adults with spine problems, 1997-2006. Spine (Phila Pa 1976). 2009;34(19):2077-2084.
- Ballantyne JC. Avoiding opioid analgesics for treatment of chronic low back pain. JAMA. 2016;315(22):2459-2460.
- Welch WP, Miller ME, Welch HG, Fisher ES, Wennberg JE. Geographic variation in expenditures for physicians’ services in the United States. N Engl J Med. 1993;328(9):621-627.
- Martell BA, O’Connor PG, Kerns RD, et al. Systematic review: opioid treatment for chronic back pain: prevalence, efficacy, and association with addiction. Ann Intern Med. 2007;146(2):116-127.
- Kobus AM, Smith DH, Morasco BJ, et al. Correlates of higher-dose opioid medication use for low back pain in primary care. J Pain. 2012;13(11):1131-1138.
- Stover BD, Turner JA, Franklin G, et al. Factors associated with early opioid prescription among workers with low back injuries. J Pain. 2006;7(10):718-725.
- Jamison RN, Edwards RR, Liu X, et al. Relationship of negative affect and outcome of an opioid therapy trial among low back pain patients. Pain Pract. 2013;13(3):173-181.
- Wasan AD, Michna E, Edwards RR, et al. Psychiatric comorbidity is associated prospectively with diminished opioid analgesia and increased opioid misuse in patients with chronic low back pain. Anesthesiology. 2015;123(4):861-872.
- Chou R, Deyo R, Friedly J, et al. Systemic pharmacologic therapies for low back pain: a systematic review for an American College of Physicians Clinical Practice Guideline. Ann Intern Med. 2017;166(7):480-492.
- Ballantyne JC. Opioid therapy in chronic pain. Phys Med Rehabil Clin N Am. 2015;26(2):201-218.
- Chaparro LE, Furlan AD, Deshpande A, Mailis-Gagnon A, Atlas S, Turk DC. Opioids compared to placebo or other treatments for chronic low-back pain. Cochrane Database Syst Rev. 2013;(8):CD004959.
- Abdel Shaheed C, Maher CG, Williams KA, Day R, McLachlan AJ. Efficacy, tolerability, and dose-dependent effects of opioid analgesics for low back pain: a systematic review and meta-analysis. JAMA Intern Med. 2016;176(7):958-968.
- Shah A, Hayes CJ, Martin BC. Characteristics of initial prescription episodes and likelihood of long-term opioid use - United States, 2006-2015. MMWR Morb Mortal Wkly Rep. 2017;66(10):265-269.
- Dowell D, Haegerich TM, Chou R. CDC Guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. 2016;315(15):1624-1645.
- Deyo RA, Smith DHM, Johnson ES, et al. Prescription opioids for back pain and use of medications for erectile dysfunction. Spine (Phila Pa 1976). 2013;38(11):909-915.
- Daniell HW. Opioid endocrinopathy in women consuming prescribed sustained-action opioids for control of nonmalignant pain. J Pain. 2008;9(1):28-36.
- Van Ryswyk E, Antic NA. Opioids and sleep-disordered breathing. Chest. 2016;150(4):934-944.
- Yi P, Pryzbylkowski P. Opioid induced hyperalgesia. Pain Med. 2015;16(suppl_1):S32-S36.
- Increases in Drug and Opioid Overdose Deaths — United States, 2000–2014. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6450a3.htm?s_cid=mm6450a3_w. Accessed August 14, 2017.
- Martins SS, Sampson L, Cerdá M, Galea S. Worldwide prevalence and trends in unintentional drug overdose: a systematic review of the literature. Am J Public Health. 2015;105(11):e29-e49.
- Deyo RA, Hallvik SE, Hildebran C, et al. Association between initial opioid prescribing patterns and subsequent long-term use among opioid-naïve patients: a statewide retrospective cohort study. J Gen Intern Med. 2017;32(1):21-27.
- Barnett ML, Olenski AR, Jena AB. Opioid-prescribing patterns of emergency physicians and risk of long-term use. N Engl J Med. 2017;376(7):663-673.