Seiji Ohtori, Yasuchika Aoki and Sumihisa Orita
Specimens from humans and animal models are known to have revealed sensory innervation of lumbar intervertebral discs and sensory nerve ingrowth into the inner layer of intervertebral discs under painful conditions. Proinflammatory cytokines, such as tumor necrosis factor-a and interleukins induce this abnormal nerve ingrowth. Nerve growth factor (NGF) has also been identified as an inducer of ingrowth, and clinical trials using NGF inhibitor are ongoing. Finally, disc degeneration induces several collagenases; their actions result in hypermobility and pain. To understand discogenic lower back pain further, the purpose of this chapter is to review pain generators and pathways of degenerative disc disease from studies of humans and animal models.
SENSORY INNERVATION OF INTERVERTEBRAL DISCS UNDER NORMAL AND PAINFUL CONDITIONS
Distribution of Nerve Fibers in Intervertebral Discs
Sensory nerve fibers innervating discs derive mainly from the sinuvertebral nerve and distribute mainly to the outer third of the disc, and their pain thresholds are relatively high. These facts suggest that intervertebral discs are relatively insensitive to mechanical stimuli under normal conditions. However, it is well known that nerve fibers with a small diameter expressing pain-related neuropeptides such as substance-P (SP) and calcitonin gene-related peptide (CGRP) are present in the sensory nerve fibers around discs. Because such fibers are generally thought to act as nociceptors transmitting pain sensation, the lumbar intervertebral disc is recognized as a source of back pain.
In 1970, Shinohara first found that nerve fibers were present in the inner part of degenerated lumbar intervertebral discs of patients with discogenic pain.1 Freemont et al. reported the presence of nerve fibers expressing growth-associated protein (GAP)-43 in inner portions of symptomatic discs.2 Because GAP-43 is recognized as a marker of axonal growth, this indicates nerve growth into the disc under pathological conditions, and this abnormal nerve ingrowth can transmit the sensation of pain from the degenerated disc. In normal discs, a tight collagen network and aggrecan-rich environment function as barriers to prevent nerve fiber growth into the disc. An annular rupture in degenerated discs disrupts the tight collagen network and induces an inflammatory response and a decrease of the proteoglycan content in the disc. Among various inflammatory mediators present in the disc, nerve growth factor (NGF) is recognized as a key factor influencing nerve ingrowth into the disc.3,4 Upregulation of NGF in the ruptured discs has already been shown in an animal model and in humans,5 implying that NGF in the ruptured disc acts as a factor promoting nerve growth into the disc.
Aggrecan, a proteoglycan found in the disc, inhibits nerve fiber growth in vitro.6 From these observations, nerve growth into the disc is triggered by an annular rupture that disrupts the tight collagen network and is promoted by an increase of inflammatory mediators and the decrease of proteoglycans, both of which occur through the process of disc degeneration.
A degenerated disc is unable to spread the load evenly over the vertebral endplate, resulting in stress concentrations on the annulus and endplate. Such abnormal mechanical stresses may stimulate the nerve fibers to grow into the degenerated disc, although at this moment for various technical reasons, there is no direct evidence showing that nerve fibers transmit pain sensation from the disc in humans.
Characteristics of Sensory Neurons Innervating the Disc
Sensory neurons innervating the lumbar intervertebral disc are present in dorsal root ganglia (DRG). DRG neurons are classified as small and large neurons. Most small DRG neurons are considered to be nociceptive and are subclassified as NGF-sensitive, and glial cell line-derived neurotrophic factor (GDNF)-sensitive neurons. This differential sensitivity to neurotrophic factors suggests that the two types of neurons have different characteristics. Almost all of the disc-innervating DRG neurons are NGF-sensitive,3 suggesting that pain sensation from the disc is transmitted mainly via NGF-sensitive DRG neurons. The distribution of the sensory fibers varies depending on the tissue. For instance, the cutaneous tissue is innervated by GDNF-sensitive neurons and NGF-sensitive neurons.3 This innervation implies that the intervertebral disc has an extremely high sensitivity to NGF because of a large amount of NGF-sensitive DRG neurons. This differential sensitivity to neurotrophic factors may explain the difference in characteristics of the pain sensation from the disc and cutaneous tissues. By contrast, the sensory neurons innervating muscles, knee joints, and visceral tissues show similar sensitivity to disc-innervating DRG neurons.7 Thus, the characteristics of discogenic pain seem more closely to resemble those of other musculoskeletal tissues such as muscle, knee joint, and visceral pain, rather than cutaneous tissue pain.
Various types of nerve fibers are present in the lumbar intervertebral disc as reviewed by Ohtori et al.8 In addition to nociceptive (SP- and CGRP-positive) and proprioceptive neurons, sympathetic neurons innervate the disc, suggesting that the pathology of discogenic pain can be affected by the sympathetic nervous system.8 Chronic discogenic pain is often influenced by psychological factors and visceral pain, such as psychosomatic abdominal pain. Involvement of the sympathetic nervous system may explain the unique characteristics of chronic discogenic pain.
In the painful discs, inflammatory mediators including NGF are upregulated9 and may affect the activity of sensory neurons innervating the discs. Yamauchi et al. reported that conditioned medium from cultured human degenerated discs upregulates SP in cocultured DRG neurons.4 They also demonstrated that SP upregulation can be blocked by an anti-NGF agent. Orita et al. reported that direct intradiscal application of anti-NGF antibody suppressed CGRP expression in DRG neurons innervating the disc.10 These findings led us to conclude that NGF can modulate the pain sensation in degenerative disc disease.
The lumbar intervertebral disc originally has a high threshold to mechanical stimuli. However, in degenerative inflammatory states, the decreased excitable threshold of sensory neurons innervating the disc in some cases may result in pathological pain from the lumbar intervertebral disc.
THE SENSORY PATHWAY FROM INTERVERTEBRAL DISCS TO DORSAL ROOT GANGLIONS (GANGLIA)
Senory Innervation of the Intervertebral Disc under Normal Conditions
The vertebra, disc, facet joint, posterior longitudinal ligament (PLL), and dura mater are innervated segmentally by the dorsal ramus and the sinuvertebral nerves branching from the spinal nerve of the corresponding levels. There are sensory nerve endings in the annulus fibrosus of intervertebral discs. It is believed that such nerve endings originate from the sinuvertebral nerves branching from the ventral ramus of the spinal nerve and the ramus communicans (RC) of the corresponding level. However, the level of the spinal cord or DRG innervating the intervertebral disc has not been fully elucidated.
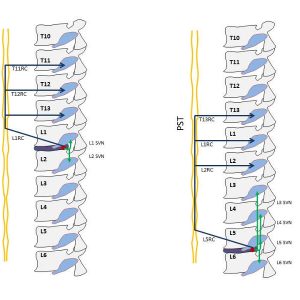
Using evidence from a rodent model, Takahashi et al. reported direct evidence for groin pain corresponding to the L2 dermatome referred from L4-5 intervertebral discs. Their experiments showed that administration of capsaicin into the anterior portion of a L4-5 lumbar intervertebral disc of rats pretreated with Evans blue (intravenously) caused dye extravasation in the groin skin corresponding to the L2 dermatome suggesting the presence of dichotomizing sensory C-fibers that innervate both the intervertebral discs and the groin skin.11 Morinaga et al. reported that the anterior portion of L5-6 discs (corresponding to the L4-5 disc in humans) were innervated by L1 and L2 DRG neurons in rats, and Nakamura et al. reported that the dense nerve network on the posterior portion of lumbar intervertebral discs had disappeared almost completely after total resection of bilateral sympathetic trunks at the level of L2–L6.12 Ohtori et al. reported “dual innervation of intervertebral discs”: that is, the dorsal portion of the L5-6 disc of rats was shown to be innervated multisegmentally by the T13 to L6 DRGs. The sensory fibers from T13, L1, and L2 DRGs were shown to innervate the dorsal portion of the L5-6 disc through the paravertebral sympathetic trunks. By contrast, those from the L3–L6 DRGs innervate the dorsal portion of the L5-6 disc through the sinuvertebral nerves.13 The dorsal portion of the lumbar discs from L1-2 to L4-5 is multisegmentally innervated by the T11 through L5 DRGs.14 Sensory fibers from the upper DRGs innervate the dorsal portion of the discs via the paravertebral sympathetic trunks, although those from the lower DRGs innervate via the sinuvertebral nerves.14 Furthermore, sensory nerve fibers enter the paravertebral sympathetic trunks through the corresponding ramus communicans and reach the DRGs via each ramus communicans at the L2 or more cranial levels, or both (Table 4-1 and Fig.4-1).
| Level of Disc | DRGs Via Sinuvertebral Nerves on PLL* | DRGs Via Paravertebral Sympathetic Trunks† |
Number of DRGs Innervating the Disc |
|---|---|---|---|
| L1/2 | L1, L2 | T11, T12, T13 | 5 |
| L2/3 | L2, L3 | T12, T13, L1 | 5 |
| L3/4 | L3, L4 | T12, T13, L1, L2 | 6 |
| L4/5 | L3, L4, L5 | T13, L1, L2 | 6 |
| L5/6 | L3, L4, L5, L6 | T13, L1, L2 | 7 |
Change of Sensory Innervation of Intervertebral Disc under Painful Conditions
The presence of SP- and CGRP-immunoreactive (ir) nerve fibers has been demonstrated in the lumbar intervertebral discs. The percentage of SP-ir T13 to L6 DRG neurons was 30%, and that of CGRP-ir neurons was 47%. The proportions of SP- or CGRP-ir neurons in L1 and L2 DRGs innervating the L5-6 disc were not different from those in L3, L4, or L5 DRGs.15 These findings indicate that under physiological conditions in rats, DRG neurons at all levels have equivalent roles in disc pain sensation. However, the ratio of CGRP-ir DRG neurons was significantly higher in L1 and L2 DRGs innervating inflamed discs than in normal controls. Associated with the inflammation in the intervertebral discs, the change in CGRP-ir neuron distribution may complicate the mechanisms of inflamed disc nociception.
Clinical Evidence of Multilevel Sensory Innervation of Intervertebral Discs
Patients who have degenerated lumbar discs in lower segments (L4-L5 or L5-S1) occasionally report groin pain. Yukawa et al. found that of 512 patients with singular lower lumbar disc herniation (L4-5 and L5-S1)21 4.1% reported groin pain and concluded that the sinuvertebral nerve that innervates the posterior annulus fibrosus, the posterior longitudinal ligament, and the dura is the afferent nerve for groin pain. Nakamura et al. reported the efficacy of an L2 spinal nerve block for patients with discogenic pain. They performed selective local anesthesia of this L2 spinal nerve for 33 patients with discogenic lower back pain. Low-back pain disappeared or significantly decreased in all patients after the L2 nerve block.12 Murata et al. also evaluated the effectiveness of the L2 block; they randomized 246 patients with lower back pain to an L2 block (n = 122) or a control block (n = 124) group. The average pain values at 5 min (P < 0.0001) and 7 days (P < 0.0001) after the block were significantly lower in the L2 block group than in the control block group.16 Ohtori et al. also reported the efficacy of the L2 spinal nerve block for patients with discogenic pain. They categorized patients into those with discogenic lower back pain and those with nondiscogenic lower back pain and examined the effect of L2 spinal nerve block in both groups. L2 spinal nerve infiltration was more effective in patients with discogenic lower back pain (21 patients, 84%) than in those with nondiscogenic lower back pain (6 patients, 40%).16 Oikawa et al. reported discogenic groin pain without lower back pain or radicular pain.17 Of 212 patients with groin pain, but without lower back pain, 5 were selected. The patients suffering from groin pain showed disc degeneration at only 1 level on magnetic resonance imaging. Patients did not show any hip joint abnormality on radiography or magnetic resonance imaging. After anterior lumbar interbody fusion surgery, groin pain significantly decreased. These results suggest that the lower intervertebral disc is innervated by DRGs at the corresponding level and multisegmentally by DRGs at upper levels.
PAIN GENERATORS IN DEGENERATIVE DISC DISEASE (DDD)
Degenerated discs contain proinflammatory cytokines such as tumor necrosis factor (TNF)-a, interleukin (IL)-6, and neurotrophins such as NGF and brain-derived neurotrophic factor (BDNF). These peptides play an important role as pain generators in DDD. TNF-a plays a crucial role in inflammation in injured degenerated discs, because it induces the release of inflammatory neuropeptides including SP and CGRP from peripheral terminals via activation of the transcription factor nuclear factor-kappa B (NF-kB) to regulate proinflammatory cytokine gene expression and the transfer of nociceptive information.18 IL-6 acts as a proinflammatory cytokine that may lead to secondary injury, and a multifunctional cytokine crucial for cell differentiation and proliferation, maintaining homeostasis, immunity, and macrophage function. IL-6 binds to membrane-bound IL-6 receptors (IL-6R). The IL-6/IL-6R complex activates glycoprotein (gp)130, initiating an IL-6 intracellular signaling cascade followed by the activation of the Janus kinase-signal transducer and activator of transcription 3 (JAK-STAT3) signaling pathway to promote inflammation and tissue injury. In the injured degenerated discs in rats, both IL-6 and its receptor are strongly expressed in the annulus fibrosus and end plate cartilage cells of injured degenerated discs in rats,19 while other inflammatory cytokines tend to exist in the nucleus pulposus cells to lead to degeneration and pain by inhibiting the synthesis of proteoglycans.20 The IL6-expressing cells infiltrating the annulus fibrosus and endplates are probably macrophages.19
Neurotrophins produced in the local site of degenerated discs are considered to induce pain-related neuropeptides in the DRG neurons. NGF produced at inflamed or degenerative sites is a key peptide involved in certain chronic inflammatory or neuropathic pain states. NGF acts by binding with two structurally unrelated receptors: tropomyosin-related kinase A (TrkA) and p75 neurotrophic receptor (p75NTR). TrkA is a specific receptor for NGF, while p75NTR has low affinity for NGF. TrkA at the peripheral nerve terminal is involved in the production of an NGF–TrkA complex, which is retrogradely transported to effect and modulate changes in pain-related gene expression in the DRG. BDNF, a neurotrophin found in degenerated discs, also plays an important role in the local inflammation together with NGF.21 Like NGF, BDNF has two structurally dependent receptors: TrkB, a specific receptor for BDNF, and p75NTR.
Some physiological changes in the DRG neurons are involved. Upregulated alteration of tetrodotoxin-sensitive voltage-gated sodium (NaV) channels, especially subunit NaV1.7, is associated with primary sensory nerve transmission in DDD.22
Some basic and clinical studies have found an analgesic effect by inhibiting/neutralizing these pain generators produced within the degenerative discs. Horii et al. reported that intradiscal injection of a TNF-a inhibitor, eternercept, significantly suppressed the production of pain-related neuropeptides in the DRG innervating degenerated discs.23 Furthermore, inhibiting its related pathway including NF-kB is effective in suppressing pain-related neuropeptides. Intradiscal injection of inhibitors for NGF, TrkA, TrkB, and p75NTR decreased production of CGRP.10,21 These basic studies have been translated into clinical studies. Some clinical studies have shown that intradiscal injection of TNF-a and IL-6 led to significant pain relief in patients with discogenic lower back pain.24,25 The intradiscal injection of inhibitors for the regular treatment for discogenic lower back pain warrants further investigation. In any event, inhibiting intradiscal proinflammatory cytokines should be a feasible option for future treatment.
REFERENCES
- Shinohara H. A study on lumbar disc lesion. Significance of histology of free nerve endings in lumbar discs. The Journal of Japanese Orthopedic Association (Japanese). 1970;44:553-570.
- Freemont AJ, Peacock TE, Goupille P, Hoyland JA, O'Brien J, Jayson MI. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet. 1997;350(9072):178-181.
- Aoki Y, Takahashi Y, Ohtori S, Moriya H, Takahashi K. Distribution and immunocytochemical characterization of dorsal root ganglion neurons innervating the lumbar intervertebral disc in rats: a review. Life Sci. 2004;74(21):2627-2642.
- Yamauchi K, Inoue G, Koshi T, et al. Nerve growth factor of cultured medium extracted from human degenerative nucleus pulposus promotes sensory nerve growth and induces substance p in vitro. Spine (Phila Pa 1976). 2009;34(21):2263-2269.
- Miyagi M, Ishikawa T, Orita S, et al. Disk injury in rats produces persistent increases in pain-related neuropeptides in dorsal root ganglia and spinal cord glia but only transient increases in inflammatory mediators: pathomechanism of chronic diskogenic low back pain. Spine (Phila Pa 1976). 2011;36(26):2260-2266.
- Johnson WE, Caterson B, Eisenstein SM, Hynds DL, Snow DM, Roberts S. Human intervertebral disc aggrecan inhibits nerve growth in vitro. Arthritis Rheum. 2002;46(10):2658-2664.
- Orita S, Ishikawa T, Miyagi M, et al. Pain-related sensory innervation in monoiodoacetate-induced osteoarthritis in rat knees that gradually develops neuronal injury in addition to inflammatory pain. BMC Musculoskelet Disord. 2011;12:134.
- Ohtori S, Inoue G, Miyagi M, Takahashi K. Pathomechanisms of discogenic low back pain in humans and animal models. Spine J. 2015;15(6):1347-1355.
- Freemont AJ, Watkins A, Le Maitre C, et al. Nerve growth factor expression and innervation of the painful intervertebral disc. J Pathol. 2002;197(3):286-292.
- Orita S, Ohtori S, Nagata M, et al. Inhibiting nerve growth factor or its receptors downregulates calcitonin gene-related peptide expression in rat lumbar dorsal root ganglia innervating injured intervertebral discs. J Orthop Res. 2010;28(12):1614-1620.
- Takahashi Y, Nakajima Y, Sakamoto T, Moriya H, Takahashi K. Capsaicin applied to rat lumbar intervertebral disc causes extravasation in the groin skin: a possible mechanism of referred pain of the intervertebral disc. Neurosci Lett. 1993;161(1):1-3.
- Nakamura SI, Takahashi K, Takahashi Y, Yamagata M, Moriya H. The afferent pathways of discogenic low-back pain. Evaluation of L2 spinal nerve infiltration. J Bone Joint Surg Br. 1996;78(4):606-612.
- Ohtori S, Takahashi Y, Takahashi K, et al. Sensory innervation of the dorsal portion of the lumbar intervertebral disc in rats. Spine (Phila Pa 1976). 1999;24(22):2295-2299.
- Ohtori S, Takahashi K, Chiba T, Yamagata M, Sameda H, Moriya H. Sensory innervation of the dorsal portion of the lumbar intervertebral discs in rats. Spine (Phila Pa 1976). 2001;26(8):946-950.
- Ohtori S, Takahashi K, Chiba T, Yamagata M, Sameda H, Moriya H. Substance P and calcitonin gene-related peptide immunoreactive sensory DRG neurons innervating the lumbar intervertebral discs in rats. Ann Anat. 2002;184(3):235-240.
- Murata Y, Kato Y, Miyamoto K, Takahashi K. Clinical study of low back pain and radicular pain pathways by using l2 spinal nerve root infiltration: a randomized, controlled, clinical trial. Spine (Phila Pa 1976). 2009;34(19):2008-2013.
- Oikawa Y, Ohtori S, Koshi T, et al. Lumbar disc degeneration induces persistent groin pain. Spine (Phila Pa 1976). 2012;37(2):114-118.
- Suzuki M, Inoue G, Gemba T, et al. Nuclear factor-kappa B decoy suppresses nerve injury and improves mechanical allodynia and thermal hyperalgesia in a rat lumbar disc herniation model. Eur Spine J. 2009;18(7):1001-1007.
- Sainoh T, Orita S, Miyagi M, et al. Interleukin-6 and interleukin-6 receptor expression, localization, and involvement in pain-sensing neuron activation in a mouse intervertebral disc injury model. J Orthop Res. 2015;33(10):1508-1514.
- Goupille P, Jayson MI, Valat JP, Freemont AJ. Matrix metalloproteinases: the clue to intervertebral disc degeneration? Spine (Phila Pa 1976). 1998;23(14):1612-1626.
- Orita S, Eguchi Y, Kamoda H, et al. Brain-derived neurotrophic factor inhibition at the punctured intervertebral disc downregulates the production of calcitonin gene-related peptide in dorsal root ganglia in rats. Spine (Phila Pa 1976). 2011;36(21):1737-1743.
- Sadamasu A, Sakuma Y, Suzuki M, et al. Upregulation of NaV1.7 in dorsal root ganglia after intervertebral disc injury in rats. Spine (Phila Pa 1976). 2014;39(7):E421-426.
- Horii M, Orita S, Nagata M, et al. Direct application of the tumor necrosis factor-a inhibitor, etanercept, into a punctured intervertebral disc decreases calcitonin gene-related peptide expression in rat dorsal root ganglion neurons. Spine (Phila Pa 1976). 2011;36(2):E80-85.
- Sainoh T, Orita S, Miyagi M, et al. Single intradiscal injection of the interleukin-6 receptor antibody tocilizumab provides short-term relief of discogenic low back pain; prospective comparative cohort study. J Orthop Sci. 2016;21(1):2-6.
- Sainoh T, Orita S, Miyagi M, et al. Single intradiscal administration of the tumor necrosis factor-alpha inhibitor, etanercept, for patients with discogenic low back pain. Pain Med. 2016;17(1):40-45.