Erfanul Saker and R. Shane Tubbs
INTRODUCTION
One of the major public health problems affecting Western industrialized societies is back pain, causing more global disability than any other condition worldwide.1 It places an enormous socioeconomic burden on the individual and health care system due to costs related to loss in working hours as well as for its medical treatment.2 In the United States, the direct annual medical expenditure on lower back pain exceeds $30 billion, which is in excess of the costs of coronary artery disease and the total costs of stroke, respiratory infection, rheumatoid disease and diabetes combined.3 In the United Kingdom, the total cost is estimated at €12 billion per annum and in the Netherlands, 1.7% of the gross national product.4 Upwards of 80% of people may experience lower back pain at some point in their life, with prevalence ranging from 15 to 45%, with around 10% of patients becoming chronically disabled.2,5 Chronic back pain is strongly associated with degeneration of the intervertebral disc.4,5,6
The normal human spinal column contains 23 fibrocartilage intervertebral discs (6 cervical, 12 thoracic, and 5 in the lumbar region), each a few millimeters thick, lying between adjacent surfaces of the vertebrae and making up roughly a quarter7 to a third4 of the height of the column. Some variations are seen in the structure between the cervical, thoracic and lumbar regions; nonetheless, the basic anatomy of the disc is similar throughout the spinal column.8 Macroscopically, the disc is divided into a peripheral fibrous cartilage called the annulus fibrosus, surrounding a centrally located gelatinous core known as the nucleus pulposus.9 The nucleus pulposus is coated inferiorly and superiorly by cartilage endplates. The intervertebral discs function to withstand biomechanical forces, provide tensile strength, allow movement (flexion, extension, and rotation) to the otherwise rigid vertebral column, and ultimately provide protection to the spinal nerves, brain and the vertebrae themselves.3,9
Intervertebral discs degenerate far earlier than other musculoskeletal tissues. This can be observed in the lumbar discs in those as young as age 11.4,5 Disc degeneration, the biochemical hallmark being the loss of proteoglycans from the disc,3 gradually increases with age, especially in males, so that around 10% of 50-year-old discs and 60% of 70-year-old discs are severely degenerated.4,5 A basic understanding of the normal intervertebral disc developmental process, anatomy, and biochemistry is of importance to gain perspective on the function and disease conditions associated with the spine and changes that arise during degeneration.
EMBRYOLOGY
The embryology of the intervertebral disc begins during the fourth week of development with the vertebral arches. These arise from sclerotome fragments of somites, which are derived from the paraxial mesoderm. Sclerotomes are defined by Sonic Hedgehog (Shh) signals secreted by the ventral floor plate and the notochord.10 The dorsal portion of the sclerotomes migrates medially around the spinal cord and notochord in a cranio-cephalic pattern to fuse with neighboring cells of the neural tube (resegmentation) to form each vertebra. Often during rearrangement of fragmented sclerotomes, it is common to have two consecutive vertebrae fuse asymmetrically or have one part of the vertebrae absent, a cause of congenital scoliosis.11 Between the cephalic and caudal parts of the segmental sclerotomes are mesenchymal cells, which during the fifth week of development fill the space between two pre-cartilaginous vertebral bodies, contributing to the formation of the intervertebral disc.11
In the region of the vertebral bodies, the notochord begins to regress and segregate along the anterior-posterior axis, eventually enlarging and contributing to the nucleus pulposus, composed of notochordal and large vacuolated cells.3 The origin of these large vacuolated cells remained ambiguous until lineage-tracing experiments using Shhcre, ShhcreERT2, and Noto-cre mouse lines elucidated the theory of notochord cells giving rise to the nucleus pulposus.3 In humans, notochord cells are maintained throughout childhood but undergo necrosis with maturation (after the third/fourth year of life) and are replaced by chondrocyte-like cells.2,9,12 Furthermore, these chondrocyte-like cells have been identified in adult nucleus pulposus in degenerated discs.3 These findings have important implication as the decrease in notochord cells and an increase in chondrocyte-like cells in the nucleus pulposus has been associated with the development of degenerative disc disease.3
As development progresses, the embryonic nuclei pulposi are later surrounded by circularly arranged fibers of the annulus fibrosus, developed from remnants of sclerotomal cells during resegmentation.11,13 These sclerotomes are initially distributed evenly, but as they migrate around the notochord, they give rise to a metameric pattern of various cell condensation.3 The annulus fibrosis is formed from dense condensation regions while the vertebral bodies form from the less condensed segments. It is speculated that Pax gene expression, in particular Pax1 and Pax9, and TGFβ-3 may be responsible for the delineation of the condensation segments.9 Important to the developing annulus fibrosus is the localization of molecular protein Noggin until birth.9 Released from the notochord, Noggin works synergistically to Shh to block the bone morphogenetic protein (BMP) signaling arising from the vertebral bodies.9
The combination of the annulus fibrosus and the nucleus pulposus constitutes the intervertebral disc between the vertebral bodies at segmental levels.11,13 By the end of the tenth week of development, the vertebral bodies are composed of cartilage (chondrocyte) cells and ossification commences.7,9 Expression of Sox genes, specifically Sox5, Sox6, and Sox9 have been implicated in chondrogenesis demonstrating the multitude of molecular signaling involved in the development of the intervertebral disc.9
ANATOMY
Normal Disc
The intervertebral disc is a heterogeneous multicomponent structure occupying approximately 25-33% (Fig. 1-1) of the spinal column’s length.4,5,7 In the vertical position, the disc confers the form of a wedge, causing the convex curvature of the lumbar spine (lordosis). Situated in spaces between adjacent vertebral bodies, apart from the atlas (C1), axis (C2) and the coccyx, each intervertebral disc consists of three basic components: a centrally positioned nucleus pulposus, two cartilaginous endplates which flank the nucleus pulposus inferiorly and superiorly, and a peripheral annulus fibrosis surrounding it (Fig. 1-2).4,5,12 The viscosity of the nucleus permits equal transmission of forces in all directions while the surrounding annulus allows the nucleus to absorb shock and dissipate axial forces.14 Although the nucleus pulposus and annulus fibrosis have distinct features relative to their location, no clear boundary exists between the nucleus and the annulus within the disc. Instead, the peripheral regions of the nucleus pulposus merge with the deeper components of the annulus fibrosis (Fig. 1-2).12 However, in younger people (<10 years), the line of delineation between the nucleus pulposus and annulus is often recognizable, becoming more vague and homogenous in adulthood.4,5,7
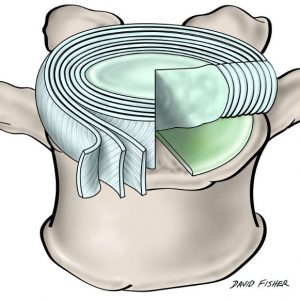
The third morphologically distinct region is the cartilaginous vertebral endplate that separates the disc from the neighboring vertebral bodies.12 It is a thin horizontal layer, usually less than 1 mm thick, containing collagen fibers oriented horizontally and parallel to the vertebral bodies, and eventually become continuous with the disc. Collagen is essential to the disc as it provides tensile strength and anchors the tissue to the bone.4,5
Structure and Composition of the Nucleus Pulposus
In the intervertebral discs of young healthy adults, the typical nucleus pulposus consists of a semi-fluid mass of mucoprotein ground substance, occupying approximately 40% of the disc’s cross-sectional area.7,12 The nucleus is centrally located and usually situated between the middle and posterior thirds of the disc.7 Histologically, it contains a few chondrocyte-like cells distributed at a low density (approximately 3000-5000/mm3), randomly organized collagen fibers, and radially arranged elastin fibers (on occasion up to 150 μm in length) interspersed in a highly hydrated aggrecan-containing gel.4,5,9,12 Biomechanically, the fluid content of the nucleus pulposus gives it the capability to be distorted under pressure, but as a fluid, its volume resists compression.12 In the event pressure is applied from any direction, the nucleus pulposus will aim to deform and will thereby transmit the applied pressure in all directions.12
The nucleus pulposus has a high-water content, approximately 70-90%, which varies throughout the day depending on activity and ultimately decreases with age.7,12 The next major component is the proteoglycans, essential to the viscosity, constituting nearly 65% of the dry weight of the nucleus. The water content of the nucleus is embedded within the domains of these proteoglycans.12 Aggrecan is the most abundant prostaglandin in the nucleus pulposus and through its fixed anionic charge, aggrecan attracts and absorbs high levels of water molecules within the nucleus. This generates a high hydrostatic swelling pressure within the irregular meshwork of collagen type II fibrils provided by its constituent chondroitin and keratan sulfate chains.4,5 This provides resistance to disc deformation and preservation of disc height.2,9 Thus aggrecan is essential for maintaining tissue hydration through the osmotic pressure. Additionally, proteoglycans contain core proteins linked to chains of glycosaminoglycans and short chains of hyaluronic acid, giving the nucleus pulposus its hydrostatic properties to allow for compression and bracing by the annulus fibrosus.15
Often the intervertebral disc is compared to articular cartilage. The distinction between the two can be made by looking at the composition and structure of the aggrecan. Aggrecan in the intervertebral disc is more highly substituted with keratan sulfate than that found in the deep zone of articular cartilage. Further, the aggrecan molecules are less aggregated (30%) and more heterogeneous in the disc, containing smaller, more degraded fragments than in articular cartilage (80% aggregated).4,5
The majority of proteoglycans in the nucleus pulposus are in the form of freely scattered proteoglycan units while only 25% are formed in aggregates.12 The proteoglycan aggregates are held in place by thin type II collagen fibrils that are distributed throughout the proteoglycan medium. The proteoglycan to collagen ratio is roughly 27:1.9 The combination of proteoglycan units, aggregates and collagen fibrils within the nucleus pulposus is collectively known as the matrix of the nucleus.12 With aging, the intervertebral disc proteoglycans become increasingly difficult to extract from the matrix.4,5
In humans with a healthy disc, the matrix goes through a continual synthesis and degradation process that determines the quality and integrity of the matrix, and thus the mechanical behavior of the disc itself.4,5 The synthesis and increased production of the matrix is attributed to the inhibition of matrix metalloproteinases (MMPs), proteolytic enzymes involved in the removal of old compartments, by fibroblast growth factor, transforming growth factor, and insulin-like growth factor.15 Degradation is facilitated by macrophages within the matrix that secrete cytokines such as interleukin (IL)-1, interferon, and tumor necrosis factor (TNF-α), which stimulate chondrocytes to secrete matrix metalloproteinases.15 In the nucleus pulposus, TNF-α has been found to activate cell migration, increase vascular permeability, and reduce matrix synthesis. Furthermore, cytokine expression studies detected increased levels of TNF-α and IL-8 in tissues of herniated disc, as well as increased vascular endothelial growth factor and nerve growth factor in degenerated discs.15
The predominant types of collagen found in the intervertebral disc are types I and II, with other types of collagen found less frequently. Collagen compromises 15-20% of the dry weight of the nucleus pulposus, and the remainder consists of radially placed elastic fibers and small quantities of noncollagenous proteins synthesized by cartilage cells.12,15 These cells are located principally in the regions of the vertebral endplates and are responsible for the synthesis of proteoglycans and collagen constituting the nucleus pulposus. Less often, type III collagen that occurs in the intervertebral disc is characteristically located near the cells of the nucleus pulposus and the inner annulus fibrosus.12
Structure and Composition of the Annulus Fibrosus
The annulus fibrosus forms the outer boundary of the intervertebral disc, enclosing the nucleus pulposus.7 It is a strong radial tire–like structure containing fibroblast cells that constantly synthesize type I and II collagen into obliquely and perpendicularly oriented lamellar fibers, forming a series of 10-20 concentric rings, or lamellae.4,5,15 Within each lamella, the collagen fibers lie parallel to one another at approximately 65° to the vertical axis between vertebral bodies, alternating to the left and right of it in adjacent lamellae (Fig. 1-2).4,5,9,12 Lamellae are interconnected through a bundle of smaller fibers consisting of fibrillin, elastin, aggrecan, lubricin and type VI collagen.9,15 Lubricin, a mucinous glycoprotein that produces lubrication within diarthrodial joints, is presumably involved in the reduction of friction between contiguous lamellae.9
The lamellae are thicker toward the center, anterior and lateral portions of the annulus; however, posteriorly they are finer and more tightly packed.12 Subsequently, the posterior portion of the annulus fibrosis is thinner compared to the rest of the annulus, which may be one of the factors attributing to the preponderance of posterior protrusion of the nucleus pulposus.7,12 Overall, the annulus is relatively stiff, providing greater strength to the intervertebral disc and allowing it to withstand compressive forces. With aging, the lamellae increase in thickness and their organization becomes progressively complex with more bifurcation and interdigitations.9
Between the lamellae are elastin fibers that constitute 2% of the annulus fibrosus dry weight. They are arranged circularly, obliquely, and vertically, passing radially from one lamella to the next while binding them together and possibly assisting the intervertebral disc in returning to its original arrangement following flexion or extension.9,12 These elastin fibers appear to be concentrated near the attachment sites of the annulus fibrosus with the vertebral endplate.12 With aging, the annulus loses its elasticity and pliability from the stress and strain placed on it.14 This has clinical implications as small fissures may form in weakened annulus regions from loss of the interwoven collagen fibers resulting in tearing, extrusion of parts of the nucleus, and abnormal displacement of the disc content against neural structures.14
The annulus can be divided into two distinct layers — the inner and the outer annulus fibrosus. The inner layer, also referred to as the transition zone, contains poorly structured extracellular matrix composed of type II collagen, proteoglycans, and water.9 Morphologically, the cells of the inner annulus are oval shaped. In contrast, the outer annulus layer is highly structured, rich in type I collagen with scant levels of type II collagen and proteoglycans, and higher resistance to tension.9 The cells of the outer layer are morphologically different as well, appearing elongated, thin, fibroblast-like, and aligned parallel to the collagen fibers.9 Cells of both the nucleus pulposus and annulus fibrosus (approximately 9000 cells/mm3) can have multiple long, thin cytoplasmic projections, some as long as or great than 30 mm long. Although the function of these projections in the disc remains obscure, it is hypothesized that they act as sensors and communicators of mechanical strain.4,5,9
The predominant structural component of the annulus fibrosus, similar to the nucleus, is water, contributing to 60-70% of its weight. The water content decreases by the time middle age is reached.16 The remaining constituents are collagen (50-60% of the dry weight) and proteoglycans, amounting to 20% of the dry weight of the annulus fibrosus.12 An estimated 50-60% of the proteoglycans of the annulus fibrosus are aggregated, mainly in the form of large aggregates.12 A degree of separation exists between the collagen fibers and separate lamellae that are packed with a proteoglycan gel, synthesized by chondrocytes (located deeper in the annulus, towards the nucleus) and fibroblasts (located principally towards the periphery of the annulus).12 This gel tethers the collagen fibers and lamellae together preventing them from buckling or fraying and also attaches the water of the annulus.12 Compared to the posterior annulus, there appears to be a greater concentration of proteoglycans and water in the anterior annulus, and in both regions increases from the outer annulus layer to the inner. Contrariwise, there are progressively fewer collagen fibrils from the outer to the inner annulus layer.12
Comparatively, the nucleus pulposus and annulus fibrosus share biochemical properties. Both have structural components composed of water, collagen, and proteoglycans.12 The only difference is in the relative concentrations of these components, specifically, the type of collagen that predominates each part. Both nucleus and annulus consist predominately of proteoglycans and water, but the nucleus only has scant type II collagen. Meanwhile, the annulus is practically ‘thickened’ by a greater concentration of collagen with type II collagen being found throughout the annulus and type I concentrated largely in the periphery of the annulus.12
Structure and Composition of the Vertebral Endplate
The anatomical intervertebral discs are bordered at the cranial and caudal ends by cartilaginous endplates, usually less than 1 mm thick, that dissect the disc and the vertebral body.9 The endplate consists of both hyaline cartilage and fibrocartilage that fully encompasses the nucleus pulposus preventing it from herniating into the adjacent vertebrae. However, in the periphery, the two endplates do not cover the annulus fibrosus in its entirety. Hyaline cartilage is present toward the vertebral body and is more prominent in neonatal and young discs. Fibrocartilage, formed by insertion of collagen fibers of the inner lamellae of the annulus fibrosus into the endplate, occurs towards the nucleus pulposus and is the main component found in older discs.12 This attachment of the annulus fibrosus to the vertebral endplates confers a strong bond to the intervertebral disc. In contrast, the endplates adhere weakly to the vertebral bodies, and can be completely torn from the vertebral bodies in certain forms of spinal trauma.12
The vertebral endplates share similar chemical characteristics with the rest of the intervertebral disc constituents. Vertebral endplates consist of high concentrations of proteoglycans, water (constituting 50-60%), and type II collagen fibers that run horizontal and parallel to the vertebral bodies, with the fibers continuing into the disc.4,5,9 Aligning along the collagen fibers are cartilage cells. Moreover, type X collagen is believed to be the essential component of endplates since it is involved in calcification and is a marker of hypertrophic chondrocytes.9 Like the nucleus and annulus, the endplates have a lower collagen content, especially towards the central region that overlies the nucleus pulposus. A reciprocal pattern is seen over the annulus fibrosus.12 The bulk of collagen across the thickness of the endplate is found in tissues nearer the bone while containing more proteoglycans and water nearer the nucleus pulposus.12 The proteoglycans are synthesized along with an extracellular matrix rich in type II collagen by chondrocytes, the only cell type constituting the endplates. A 1:2 collagen to proteoglycan ratio is observed in endplates, similar to that found in articular cartilage.9
Similar to the role proteoglycans play in the nucleus pulposus and annulus fibrosus, proteoglycans in the endplate preserve the water content through the intervertebral disc. Additionally, they are necessary for the control of solutes. The loss of proteoglycans from the endplate cartilage is associated with loss of proteoglycans from the nucleus pulposus, which has an important clinical implication; the depletion of proteoglycans would eventually lead to intervertebral disc degeneration.9 Additionally, any alterations in the biochemical makeup of the disc, especially in the endplate, during the skeletal growth may also be involved in the development of scoliosis.9
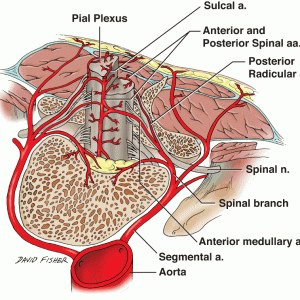
Blood Supply
The healthy adult nucleus pulposus and inner annulus fibrosus are completely avascular. However, a few blood vessels exist around the peripheral annulus. External and endplate capillaries, branching from segmental arteries, which in turn branch off the aorta, provide blood with nutrients to the region of the outer annulus fibrosus. This blood drains to the subchondral venous plexus or into the veins of the marrow spaces of the adjacent vertebral body.9
Although blood vessels may not be present in the adult disc, from as early as 35 weeks of development up to the age of eight years, blood vessels penetrate the annulus and cartilaginous endplates and are derived from the segmental vessels (Fig. 1-3).7,9 A central axial vessel, termed the marginal vessels, can be seen traversing a vertical course from the osseous vertebrae to the endplate and two additional ventral-dorsal vessels.7 During the first three decades of life, the marginal vessels are gradually obliterated leaving the cartilaginous endplate with residual scar tissue. Adjacent to the disc in young cartilaginous endplates (less than 12 months old) are blood vessels present in the longitudinal ligaments, which are branches of the spinal artery.4,5 Ultimately, as the vessels recede, completely obliterated by cessation of growth, the intervertebral disc becomes nearly avascular with very minimal vessels present in the endplate.7,9
It is believed the vascular regression may be due to decreased nutrient requirements following the initial period of rapid growth or the inadequacy of the circulatory pressure to contest the large physiological stresses in the surrounding extracellular matrix.9 Further, the paths followed by these blood vessels never fully remodeled and left translamellar bridging elements, whose function have yet to be determined. It is postulated they may respond to radial and shear deformations. However, it is debatable whether this influence would facilitate or diminish the function of the intervertebral disc.9
During the development and growth of the intervertebral disc, the endplates contain a mesh of microscopic blood vessels that are responsible for nutritional intake. Despite the resemblance to the rest of the disc, the endplate does not confer an additional barrier to diffusion.12 Rather, metabolites diffuse through pores present in the growth plates, created via marrow contact channels resultant from areas of deficiency in subchondral bone of the vertebral body.9,12 Approximately 80% of the nutrients are transported to the intervertebral disc through this route.9 Diffusion is based on the size and ionic charge of the various molecules.12 The high concentration of proteoglycans in the nucleus pulposus generates a net negative charge thus allowing only cations (e.g., sodium, calcium) or neutral molecules, such as glucose and oxygen, to diffuse from the vertebral body to the center of the disc.9,12 Negatively charged ions (e.g., sulfate, chloride) and macromolecules (e.g., immunoglobulins and enzymes) are impeded from diffusion through these channels.9
The metabolic activity of the intervertebral cells requires the various nutrients and substrates obtained through diffusion, however, the limited blood supply reaching the disc creates variations in oxygen concentration and pH level throughout the different zones of the disc.9 The oxygen tension in the center of a disc is only 2-5% of those at its periphery. Consequently, cells resort to using an anaerobic metabolism, thus producing a high concentration of lactic acid. This makes the environment of the disc acidic with a pH in the range of 6.9-7.1.12 Notochord cells found in the nucleus pulposus are sensitive to pH fluctuations. In vitro experiments have demonstrated that a chronic lack of oxygen causes these cells to become inactive, while a chronic lack of glucose leads to their death.9
Muscarinic receptors have been identified suggesting not all blood flow to the endplate region occurs via passive diffusion. These receptors may possibly influence disc nutrition under altered physiological conditions.9 By the time the network of microscopic blood vessels of the endplates reaches skeletal maturity, they mostly involute leaving residual “weak spots.” Subsequently, these may lead to Schmorl node formation and later, to sclerosis of the subchondral plate.9
Adjacent Ligaments and Innervation of the Intervertebral Discs
The anterior longitudinal ligament is a thick, strong ligament localized to the anterior and anterolateral aspects of the vertebral bodies traversing the atlas down to the sacrum. It is firmly bound to the vertebral bodies at its periphery, with the edges of the ligament being thinner than the innermost segment. The deepest fibers of this ligament merge with the intervertebral disc and extend from the body of one vertebra to the disc, to that of the neighboring vertebra. These same deep fibers attach the discs with the margins of the vertebrae.7 The anterior longitudinal ligament and adjacent disc receive afferent innervation from branches that originate in the dorsal root ganglion and anterior sympathetic plexus (Fig. 1-4).4,5
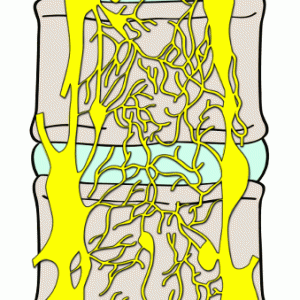
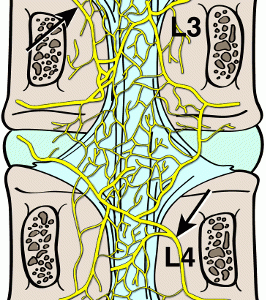
The posterior longitudinal ligament rests on the posterior surface of the vertebral bodies extending from the atlas to the sacrum. A meningeal branch of the spinal nerve, known as the recurrent sinuvertebral nerve, originates near the disc space. Nociceptive fibers from the ascending branch of the sinuvertebral nerve supply the posterior longitudinal ligament and adjacent annulus (Fig. 1-5).4,5,7 The nerve exits from the dorsal root ganglion and enters the foramen, where it divides into a major ascending and a lesser descending branch.4,5 Animal studies demonstrated further sympathetic afferent contributions to the sinuvertebral nerve arising via the rami communicantes.4,5,7 Bogduk17 and colleagues performed cadaver studies and detected innervation of the lumbar intervertebral discs by sinuvertebral nerve fibers on the outer posterior region of the annulus fibrosus.15 They hypothesized dual innervation from a sympathetic branch from the gray ramus communicans and a somatic branch from the ventral ramus.15 The sinuvertebral nerve has clinical implications as it carries many of the pertinent sensory fibers from the organs concerned with the production of low back pain.7 Additionally, evidence has shown increased sympathetic afferents in degenerating intervertebral discs.9
In the thoracic and lumbar levels, the posterior longitudinal ligament becomes narrow as it passes through the vertebral bodies and then expands over the discs, taking on the configuration of an hourglass. The lateral expansions over the intervertebral discs are weaker compared to the strong central band and thus forms a less well-enforced region for potential disc herniation.7
Anatomic studies have demonstrated an extensive network of fine free nerve fibers located on the intervertebral disc surface and relatively few nerve endings, mainly unmyelinated and complex unencapsulated, within the outer lamella of the annulus fibrosus.4,5,7,15,17,18 The immunohistochemical profile of these nerve fibers has shown to test positive for substances such as acetylcholinesterase, neurofilament protein, substance P, calcitonin gene-related peptide, vasoactive intestinal polypeptide, neuropeptide Y, C-flanking peptide and synaptophysin.9,19
Isolated studies of the cervical spine have demonstrated nerve fibers in the anterior and posterior longitudinal ligaments as well as the superficial layers of the annulus fibrosus.7 However, the deeper layers of the annulus and the nucleus pulposus were aneural. Interestingly, with progressive degeneration, the intervertebral disc becomes densely innervated even in regions that in normal conditions lack innervation, like the inner part of the annulus, and sometimes even to the nucleus pulposus.9,19 These nerves were mostly nociceptive fibers that may accompany ingrowing blood vessels.9 The mechanisms responsible for nerve growth and hyperinnervation of pathological intervertebral discs have not been fully understood. It has been theorized that neurotrophins may play a role. These neurotrophins are known to have both neurotrophic and neurotropic properties and regulate the density and distribution of nerve fibers in peripheral tissues.19 Neurotrophins and their receptors are expressed in healthy discs, but substantially higher levels have been observed in pathological discs, thus suggesting a correlation between levels of expression of neurotrophins and density of innervation in intervertebral discs.19
Additional fibers were found in the dura and periosteum surrounding the intervertebral disc.4 Therefore, any pain originating from the displacement of the intervertebral disc contents might occur related to pressure on structures carrying pain-sensitive nerve endings or fibers.7,14,20 Direct impingement on the anterior longitudinal or annulus fibrosus has also been shown to induce pain.14,20
Most of the fibers innervating the intervertebral disc are sensory nociceptive, and to a lesser extent proprioceptive. A few mechanoreceptors are present in the outer two to three lamellae of the annulus fibrosus lamellae, most frequently having the morphology resembling Golgi tendon organs, less often Ruffini endings and Pacinian corpuscles.9,19 Some of the nerves in discs also have glial support cells (Schwann cells) alongside them.4,5 Degenerating discs have been shown to have an increase in the number of Golgi-tendon organ-like structures as well as increased nerve growth factor expression.9
The healthy adult cartilaginous endplate, like other hyaline cartilages, is normally fully avascular and aneural.4,5,9
Specifics of the Lumbar Intervertebral Discs
In the lumbar region of the spine, there are five intervertebral discs, the lowest of which is situated atop the sacrum. Compared to the discs of the thoracic and cervical spine, the lumbar discs are taller and wider measuring approximately 7–10 mm in thickness and 4 cm in diameter (anterior-posterior plane).4 Morphologically, the discs are cylindrical with its shape being determined by the integrity of the annulus fibrosus.21 The lumbar discs tend to be of greater height anteriorly than posteriorly and this tendency is especially noticeable in the fifth lumbar disc. In the upper segment of the lumbar spine, the lordosis is due almost entirely to the shape of the disc, but in the lower lumbar regions, the shape of the vertebral body is a contributing factor.7
It is established that the lumbar intervertebral discs are innervated segmentally. The nerve fibers innervating the posterolateral portion of the discs recognized for being the region with the most frequent lesions in human, come from the T13-L6 dorsal root ganglion but are mainly localized in the L1-L2 dorsal ganglion neurons. It is for this reason the lumbar region is the main source of discogenic pain in humans.19 These discs are more vulnerable to injury than either the cervical or thoracic disc because they are subject to a great range of motion while under great axial load. The weight of the body and downward gravitational force generates more burden and load to the lumbar region. A similar force and axial load are placed on the thoracic region, however, the connecting rib cage mitigates much of that force and limits the range of motion.
It has been suggested the underlying mechanisms contributing to lumbar radiculopathy include local chemical irritant resulting from proteoglycan release from a disc. This release could then create an inflammatory reaction, an autoimmune reaction from exposed disc tissue, or an increased concentration of lactic acid and/or a lower pH around the spinal nerve roots.21
From L2 downward, the spinal nerves leave the dura more proximally than the foramen through which they pass, at a decreasing angle of obliquity, and with an increasing length of the canal.21 The lumbar nerves are bundled sufficiently high in the foramina such that they are unaffected by a degenerated disc at the same level unless segments of the disc migrate cranially. Therefore, the fifth lumbar nerve root will more often be compressed by an L4-L5 degenerated disc than by a degenerated L5-S1 disc.7 Roughly 40% of intervertebral disc impairments affect the fourth and fifth lumbar nerve roots.21
SUGGESTED READINGS
- Arslan M, Cömert A, Açar Hİ, et al. Nerve root to lumbar disc relationships at the intervertebral foramen from a surgical viewpoint: an anatomical study.Clin Anat. 2012;25(2):218-223.
- Arslan M, Cömert A, Açar Hİ, et al. Neurovascular structures adjacent to the lumbar intervertebral discs: an anatomical study of their morphometry and relationships. J Neurosurg Spine. 2011;14(5):630-638.
- Bailey RW, Sherk HH, Dunn EJ, et al. The Cervical Spine. Philadelphia, PA: Lippincott; 1983.
- Smith GW, Robinson RA. The treatment of certain cervical-spine disorders by anterior removal of the intervertebral disc and interbody fusion. J Bone Joint Surg Am. 1958;40(3):607-624.
REFERENCES
- Hoy D, March L, Brooks P, et al. The global burden of low back pain: estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis. 2014;73(6):968-974.
- Daly C, Ghosh P, Jenkin G, Oehme D, Goldschlager T. A review of animal models of intervertebral disc degeneration: pathophysiology, regeneration, and translation to the clinic. BioMed Res Int. 2016;2016:5952165.
- Sivakamasundari, Lufkin T. Bridging the gap: understanding embryonic intervertebral disc development. Cell Dev Biol. 2012;1(2):103.
- Urban JP, Roberts S. Degeneration of the intervertebral disc. Arthritis Res Ther. 2003;5(3):120-130.
- Raj PP. Intervertebral disc: anatomy-physiology-pathophysiology-treatment. Pain Pract. 2008;8(1):18-44.
- Bogduk N. The anatomical basis for spinal pain syndromes. J Manipulative Physiol Ther. 1995;18(9):603-605.
- DePalma AF, Rothman RH. The Intervertebral Disc. Philadelphia, PA: W.B. Saunders Company; 1970.
- Coventry MB. Anatomy of the Intervertebral Disk. Clin Orthop Relat Res. 1969;67:9-15.
- Tomaszewski KA, Saganiak K, Gładysz T, Walocha JA. The biology behind the human intervertebral disc and its endplates. Folia Morphol (Warsz). 2015;74(2):157-168.
- Ehlen HW, Buelens LA, Vortkamp A. Hedgehog signaling in skeletal development. Birth Defects Res C Embryo Today. 2006;78(3):267-279.
- Sadler TW. Langman’s Medical Embryology. 10th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2012.
- Bogduk N. Clinical Anatomy of the Lumbar Spine and Sacrum. 3rd ed. New York, NY: Churchull Livingstone; 1997.
- Larsen WJ. Human Embryology. 3rd ed. Philadelphia, PA: Churchill Livingstone; 2001.
- Camins MB, O’Leary PR. The Lumbar Spine. New York, NY: Raven Press; 1987.
- Vora AJ, Doerr KD, Wolfer LR. Functional anatomy and pathophysiology of axial low back pain: disc, posterior elements, sacroiliac joint, and associated pain generators. Phys Med Rehabil Clin N Am. 2010;21(4):679-709.
- Lang J. Clinical Anatomy of the Cervical Spine. New York: Thieme; 1993.
- Bogduk N, Tynan W, Wilson AS. The nerve supply to the human lumbar intervertebral discs. J Anat. 1981;132(Pt 1):39-56.
- Hirsch C, Ingelmark BE, Miller M. The anatomical basis for low back pain. Studies on the presence of sensory nerve endings in ligamentous, capsular and intervertebral disc structures in the human lumbar spine. Acta Orthop Scand. 1963;33:1-17.
- García-Cosamalón J, del Valle ME, Calavia MG, et al. Intervertebral disc, sensory nerves and neurotrophins: who is who in discogenic pain? J Anat. 2010;217(1):1-15.
- Compere EL. Origin, anatomy, physiology, and pathology of the intervertebral disc. Instr Course Lect. 1961;18:15-20.
- Dutton M. Orthopaedic Examination Evaluation and Intervention. 2nd ed. New York, NY: McGraw-Hill Global Education Holdings; 2008.