Peter A. Robertson
INTRODUCTION
The Failed Spine Surgery candidate for revision surgery encompasses all patients who have undergone previous spine surgery in the lumbar region. The previous experience of the patient, as well as the context within which the surgeon is seeing that patient, may be highly variable and influence expectations and approaches for both parties. The patient who has had a successful previous discectomy and presents with new pathology after a prolonged period of symptom relief will differ in approach and expectations from the patient who has suffered ongoing, persistent symptoms and disability despite recent surgery. Similarly, the treating surgeon will likely experience differing thoughts and emotions regarding an old patient who underwent successful surgery in the past and who now has a new problem, compared to a recent patient whose surgery has been unsuccessful and who comes to the clinic seeking explanations, understanding and further intervention. While a surgeon providing a second opinion gives a failed spine surgery patient a full assessment as a new patient, for a patient who has failed to meet expectations an evaluation during the limited time allocation of follow-up represents multiple challenges for the primary operator. What is clear is that when both the patient and the surgeon agree that the previous intervention has not resulted in the expected symptom relief, or when a patient has had previously successful surgery only to later develop pain and disability, a precisely structured approach to the evaluation of the patient offers the best chance to clearly identify pathologies that are responsible for ongoing symptoms. This structured approach then allows for a logical management plan with further surgical intervention based on very clear surgical goals and expectations, rather than mere hope and desperation.
To clarify approaches for Failed Spine Surgery Patients, it is useful to look at failure from the perspectives of time, symptom type and potential pathologies.
| Primary Surgery Type | Leg Pain Dominant/ Nerve Root/Radicular Pain |
Back Dominant/Axial Pain |
|---|---|---|
| Microdiscectomy/Discectomy | Wrong Level/Side Surgery Missed Prolapse Recurrent Disc Prolapse “Scar”/Radiculitis |
Discogenic Pain Facet Pain Infection/Discitis |
| Decompression for Spinal Stenosis | Inadequate Decompression/Missed Pathologies Recurrent Stenosis Spondylolisthesis Scoliosis and Foramen Stenosis Adjacent Segment Degeneration with Stenosis Arachnoiditis |
Facet Overload/ Destruction/Pain Pars Failure (Iatrogenic) Postoperative Spondylolisthesis Postoperative Scoliosis Sagittal Imbalance |
| Fusion of a Motion Segment | Nerve Root of Cauda Equina Damage Nerve Root Compression Adjacent Segment Pathologies – HNP/SS |
Inadequate Stability Interbody Implant Subsidence Nonunion Screw Loosening Adjacent Segment Disease Sagittal Imbalance |
| Motion Preservation Devices (Interspinous Spacers (ISS) and Total Disc Arthroplasty (TDA)) |
Cauda equina or Nerve Root Impingement Progressive Stenosis or HNP at index level Adjacent Segment Disease with HNP/SS |
Inadequate Stabilization (ISS) Failure to recognize Facet Symptoms (TDA) Facet Overload Deformity Progression |
Time
Failure of spine surgery should be categorized as occurring immediately, early or late. Immediate failure of surgery is obvious—the patient has no sustained relief of the symptoms for which surgery was performed. Typical examples include incorrect diagnosis or wrong level surgery. In both cases, the original symptomatic pathology will not have been treated by the surgery.
Early failure is somewhat more difficult to define, and strict timelines are unlikely to be helpful. With early failure, the patient and surgeon both agree that there has been good amelioration of symptoms for which surgery has been performed; yet some time later, generally within a year, symptoms have returned. Symptom recurrence may be gradual or sudden, associated with some event or trauma, and represent either recurrence of symptoms from the original problem or a new diagnosis. Early failure can be the most difficult to evaluate in that almost all the potential pathologies may be implicated.
Late failure after lumbar spinal surgery occurs when there has been sustained relief of index symptoms and when the patient has made a good recovery to acceptable activity levels, often over a period of many years. This can be a very positive situation for both the patient and the surgeon to approach further evaluation. The patient has a positive view of the previous intervention and its benefits, while the surgeon will likely be able to focus on new pathologies or late recurrence of neurocompressive disorders, such as recurrent disc prolapse or recurrent spinal stenosis. Oftentimes, late failure indicates new pathologies in different or ‘adjacent’ regions of the spine, with the potential for successful treatment compatible with the original operation.
Symptom Type and Potential Pathologies
Failure in terms of previous spinal surgery may also be considered in the context of symptom type. Leg dominant pain of a neuralgic character is likely related to nerve root compression within the lumbar spine, and may be contrasted with back-dominant pain due to axial pathology within the lumbar spine. In many cases, although we attempt to ascribe ‘dominance’ of symptoms, pathologies may cause both radicular (nerve root) symptoms and axial pain.
Leg dominant symptoms: Leg symptoms from radicular pain secondary to nerve root compression relate to either missed or undertreated pathology (if the failure is immediate) or new pathology (if the failure occurs later). When a patient who has a clear isolated disc prolapse fails to respond to discectomy/microdiscectomy, then consider wrong level surgery, wrong side surgery, or a missed fragment. If the patient’s leg dominant symptoms were due to spinal stenosis, then consider again incorrect-level and -side surgery before looking for incomplete decompression. Incomplete decompression results from the lateral recess of the spinal canal being inadequately decompressed by a medial facetectomy, or from unrecognized foramen pathology which causes lateral compression of the nerve root. Additionally, inadequate decompression may occur when the dynamic compression associated with upright posture during standing and walking is not appreciated from a supine MRI scan. Subtle degrees of spondylolisthesis evident on erect radiographs may be missed on the recumbent MRI, where the only clue is facet gapping. Foramen compression frequently occurs for the patient who has a subtle scoliosis. The roots are compressed in the concavity of the curve, yet the scoliotic curve will be underappreciated if the primary evaluation used supine MRI alone, without erect radiographs to assist with understanding of alignment.
Immediate failure of spinal surgery is also recognized under the label of “battered root syndrome” – an iatrogenic etiology. Blunt trauma to the nerve root is hopefully less common with precision imaging of pathology and appropriate application of surgical techniques. However, direct trauma may still be exhibited when dural tears occur indicative of trauma to the cauda equina and roots, and secondary to instrumentation misplacement. Such instrumentation misplacement, particularly pedicle screw misplacement and, in the past, sublaminar wire use, may damage nerve roots and result in troubling appearances on cross-sectional imaging, specifically CT scans. Identification of iatrogenic nerve root damage may require exclusion of all other potential causes, or it may be clear on cross sectional imaging. Its identification is of great importance, as recovery from iatrogenic nerve root damage is unpredictable; even with further intervention, prognoses must be circumspect.
Back-dominant symptoms: Back-dominant symptoms reflective of axial pain generation may have multiple potential origins. Following discectomy, the segmental biomechanics will change with the potential for a small number of patients to develop back-dominant symptoms despite good relief of leg pain. The disc and or the facet joints may be the source of symptoms.
After decompression surgery, back-dominant pain may have several origins. Underlying spondylosis may cause ongoing back pain despite adequate decompression to relieve nerve root compression. Facet failure with rapid progressive joint destruction, iatrogenic or acquired pars fracture, and development of deformity are common problems. The combination of altered biomechanics due to decompression and progression of the degenerative process can result in progressive postoperative spondylolisthesis and degenerative scoliosis. Facet and disc failure can also contribute to axial symptoms.
After fusion procedures, back-dominant pain may occur for many reasons. Nonunion is a popular presumed cause of pain after fusion, although not all nonunions would seem to be painful. Nonunion may occur after failed or inadequate stabilization, poor host factors such as nicotine smoking, and inadequate attention to bone grafting substances and techniques to achieve fusion. Fusion malunion is common, yet it is only becoming recognized in the modern era. Many disorders that result in lumbar fusion are associated with disc height loss in the lumbar spine; when that loss occurs in the low lumbar spine, loss of segmental lordosis occurs. The mobile segments above the abnormal segment compensate for the lordosis loss, but once they can no longer compensate, sagittal imbalance occurs. Fusion procedures that occur in-situ without correction and normalization of the sagittal segmental alignment will immediately require compensation at the adjacent segment, with abnormal biomechanical loading and the potential for accelerated degeneration and symptom production.
Additional pathologies require consideration when axial back pain is the dominant symptom. Deep infection in the intervertebral disc, after discectomy, is uncommon but may cause significant morbidity. Pain at rest is a key indicator. The patient may be systemically unwell, and raised inflammatory markers – the ESR and CRP – will often support typical imaging findings denoting endplate destruction and vertebral inflammation. Percutaneous biopsy may be required to optimize identification of the infective organism if blood cultures are negative.
Fractures within the pars may explain postoperative spondylolisthesis; however, vertebral body fractures are more frequent in the osteopaenic patient at the superior end of a multilevel fusion. Sacral insufficiency may result in fracture below a fusion. These pathologies must especially be considered in the elderly patient or in those with metabolic bone disease.
Further Care
The Failed Spine Surgery patient also presents challenges from the rehabilitation perspective. Prolonged pain and disability often result in muscle loss and reduced range of motion. For want of a better phrase, these patients are ‘deconditioned’ – they need to understand that no amount of reconstruction or removal of the ‘pain generator’ will transform a highly disabled and deconditioned patient into a person of normal activity for their age. Realistic expectations from all parties – and commitment to physical rehabilitation – are key requirements before any planning for revision surgery.
Finally, in terms of introductory comments, the spine community must recognize that a commitment to the evaluation and care of the Failed Spine Surgery patient is essential. The increasing incidence of surgical interventions, often occurring at a younger stage of life, within a population that has an increasing life expectancy with increased demands in terms of activity levels, means that the frequency of patients who have had previous surgery and require re-evaluation will increase for the foreseeable future. The complexity of revision surgery means that it is expensive; in an environment of cost awareness and containment, it is essential that precision in diagnosis and treatment maximize not only outcomes, but also cost effectiveness.
SURGICAL CONSIDERATIONS WHEN PLANNING REVISION SURGERY
The most common considerations for revision surgery will involve techniques for revision discectomy, revision decompression and revision fusion. Each of these topics has been fully summarized in the following chapters. When evaluating the Failed Spine Surgery patient, there are several other general considerations addressed below.
Wrong Site Surgery
With the widespread use of intraoperative imaging, wrong site surgery should be diminishing in frequency. However, pitfalls still exist, and the surgeon must take care to be extra vigilant.
Transitional anatomy at the lumbosacral junction can be confusing. Failure to recognize sacralization of L5 will influence any intraoperative counting on the basis of segment mobility. Radiologists may use numbering that differs from the operating surgeon, particularly if they count down from the last rib at the thoracolumbar junction. Ankylosis of segments may confuse again if the surgeon is counting levels based off intraoperative segmental motion. Upper lumbar interpretation of intraoperative imaging can be confusing when representing the further distance from the numbering base – the sacrum. Difficulties with level identification are particularly evident up into the thoracic spine, particularly if counting of levels is based on ribs that are assumed to be of a normal number and preoperative imaging has not established the segmentation of the thoracolumbosacral spine.
If there is any doubt regarding the segmentation of the lumbar spine, preoperative plain radiographs that confirm the presence or absence of a transitional level, as well as meticulous correlation with sagittal imaging studies (MRI), will minimize the risk of wrong level surgery. While there are a number of conventions in relation to level numbering, this author believes that the system is less relevant than the conviction of recognizing ‘difficult’ anatomy and meticulously correlating with plain radiographs or CT 3D reconstructions with a particular emphasis on axial imaging to identify the pathology level.
When performing revision surgery, the surgeon should be aware that although initial surgery may have been performed at a particular level, it is not uncommon for the primary surgeon to have explored other levels. This surgical dissection may again confuse the revising surgeon.
Revision in the presence of instrumentation should also assist in level identification. However, the distal relationship of the facet joint in relation to the proximal pedicle may cause confusion and so should be carefully considered.
Spinal Alignment/Malalignment
For a long time, correct alignment of peripheral joint and scoliotic reconstruction surgery has been recognized as a requirement for positive outcomes. However, attention to alignment in lumbar fusion surgery has been conspicuously absent. Instead, attention to surgical indications, instrumentation to achieve fusion in multilevel constructs, and bone graft types and biologics has diverted attention from the understanding of deformities and the options to correct those deformities.
The alignment in the coronal plane has been a long-term goal of scoliosis surgery, and more recently the need to optimize sagittal alignment has become clear in both adolescent and adult deformity reconstruction.1 This attention to alignment has until recently not been a focus in lumbar fusion surgery, and fusion, instrumented fusion, and ‘fusion in situ’ all fail to understand the deformities that occur within the degenerative process.
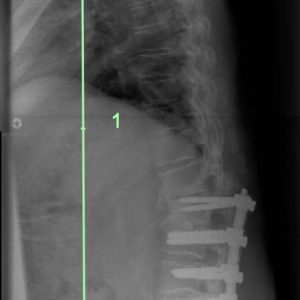
In adult deformity surgery the aim of deformity correction in the sagittal plane is to optimize sagittal balance. This is generally achieved by recreating a lumbar lordosis that matches the pelvic shape, reducing the need for deformity compensation at other (proximal) spinal segments or peripheral joints such as the hips or knees. These concepts are important and reflect the sagittal measurements that are employed in classification of adult deformity.2 Health-related quality of life outcome measures correlate loss of sagittal balance (forward stoop) with poorer outcomes (Fig. 1-1). The measure of sagittal balance is the Sagittal Vertical Axis (SVA), which measures the forward displacement of a plumb line dropped from C7 in relation to the posterior corner of the superior endplate of the sacrum. All measures are made on full length spine radiographs that include the pelvis and hip joints and are made in the erect position with the patient’s knees fully extended. Normal adults have a measure of 0–40 mm anterior displacement of the C7 plumb line. In the elderly population this measure increases. Marked increases of the SVA result in difficulty standing and walking, high energy utilization, rapid fatigue and poor function. Surgery to correct sagittal alignment requires recreation of the lumbar lordosis to match the pelvic shape.
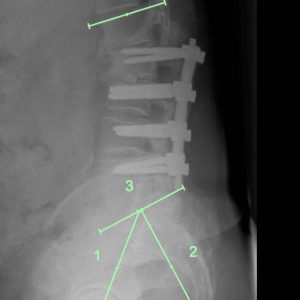
The Pelvic Incidence (PI) defines the pelvic shape in the sagittal plane. The PI is an angular measurement made by the angle between a line from the hip axis to the midpoint of the sacral endplate and a line perpendicular to the sacral endplate.3 A low PI correlates with a low sacral slope (a relatively flat sacral endplate) and therefore only requires a small lumbar lordosis (LL = the angular measurement between the superior endplate L1 and the S1 endplate) to balance the head and trunk above the pelvis. Conversely, a high PI correlates with a high sacral slope, and this in turn requires a large lumbar lordosis to achieve sagittal balance (Fig. 1-2).
The PI is interesting in that it is highly variable (range 20–90°).4 There are few other body sites where the variability in range of anatomy that would determine alignment is so large. Consequently, the normal lordosis range is also large (20–90°).
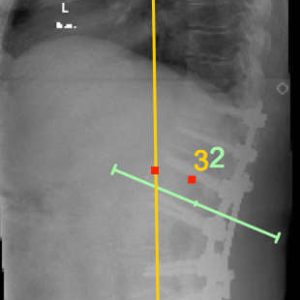
In adult reconstruction, the need to match the lordosis with the PI is denoted by the equation PI = LL (+/- 10°) (Fig. 1-3). Better clinical outcomes occur when PI = LL.5 Conversely, PI/LL Mismatch (PI ≠ LL) is associated with poor outcomes. Almost universally, the LL is much smaller than the PI, resulting in a forward stoop unless there is compensation, generally by extension at the hip joint, which brings the trunk back over the sacrum and reduces the SVA. The measure of compensation due to hip extension is the Pelvic Tilt (PT). It is calculated as the angle between the vertical and the previously utilized line between the hip axis and the midpoint of the sacral endplate.
To summarize, the key measures of sagittal alignment in adult deformity are the PI, LL, PT and SVA, which characterize the pelvic shape, the lumbar lordosis (that matches the pelvic shape in the normal individual), the presence of any compensation by hip extension (PT), and the global sagittal alignment. The importance of sagittal alignment is such that these measures – and departures from the normal measurements – dominate the Adult Deformity Classification system2 with considerably less focus on coronal plane alignment than exists in idiopathic scoliosis classification systems (Fig. 1-1).
The relevance of these concepts of sagittal alignment to lumbar reconstruction, often only over a single spinal level, is becoming increasingly clear. Recent attention to PI/LL relationships has shown that PI ≠ LL is associated with inferior outcomes for patients in terms of pain and disability,6 even across a range of diagnoses.7,8 Most importantly, it is also associated with more rapid adjacent segment degeneration and earlier required revision.9 Thus, optimization of sagittal alignment will favorably influence the outcomes and longevity of fusion surgery.
If, however, we look deeper into measures of sagittal alignment when surgery is limited to single- and double-level fusion, it is clear that the PI/LL relationship is a very broad measure only, and will miss compensation that occurs within the lumbar lordosis for lack of lordosis in any reconstructed segment. The spine will often compensate for a hypolordotic region with local hyperextension in the proximal segment or segments. A segment with hyperextension will have abnormal mechanics and may well generate pain, yet its significance will be missed with a global measure of lumbar lordosis such as the PI/LL relationship.
A deeper understanding of the anatomical contributions to lordosis is particularly valuable. In the average PI of 50–55° the lordosis will be of similar magnitude. This lordosis is made up of lordotic angulation of the vertebral bodies which is generally found mainly at L5 with a mean value of 10°. The other vertebrae are rectangular. The remaining lordosis comes from lordotic angulation of the discs, with approximate values of 3° at L1/2, 6° at L2/3, 9° at L3/4, 12° at L4/5 and 15° at L5/S1. The total lordotic angulation is thus 45° in the discs with 10° from the wedge shape of L5 – giving a lordotic angulation of 55 degrees – very similar to the PI of 50–55°.10 Now, it is essential to emphasize that these values are very much a ‘mean’ value. An individual with a PI of 20° will have lordosis with much less of a contribution from the discs and L5 shape. Conversely, an individual with a PI of 90° requires a lordosis with much greater individual lordotic angulations in the discs, sometimes in the L5 and L4 vertebral body wedging, with a greater lordotic contribution coming from the upper lumbar discs.11
There is increasing evidence that the population of individuals at the two extremes from the mean PI are at risk of certain pathologies in the lumbar spine. Low PI is related to a greater incidence of Disc Degenerative Disease,12 while high PI is associated with shear failure in the low lumbar spine – including both isthmic and degenerative spondylolisthesis.13
While there has been limited focus on the kyphosis at the lumbosacral junction in association with high-grade spondylolisthesis, there is at present little focus upon the angular deformity that is associated with common lumbar pathologies that present for reconstructive surgery. This is despite the logical conclusion, from the data on normative values for disc lordosis, that a patient with total disc height loss in association with a spondylolisthesis at L5/S1 will likely have lost 15° of contribution to the lordosis. Compensation from segments above will normalize the lordosis and avoid detection by the PI/LL metric, yet the deformity will not be corrected by any fixation in situ, likely leading to accelerated changes at the adjacent segment. While surgeons for other regions of the body would not accept uncorrected deformities of this magnitude, attention to this degree of detail within assessment and reconstruction planning in lumbar degenerative surgery does not appear to be widespread. It is also clear that the reconstruction options (TLIF/PLIF/LLIF/ALIF) vary in their ability to recreate lordosis,14 and therefore should be an increasing focus of future studies. Ideally, preoperative planning would involve a unisegmental assessment of angular deformity associated with the pathology requiring treatment, followed by operative planning that incorporates angular correction during reconstruction with fusion. For the surgeon planning revision surgery, it is increasingly evident that there is a need to understand the normal sagittal alignment specific to the individual patient’s anatomy, and then to incorporate that knowledge into any revision procedure. Recreation of appropriate alignment will become an increasing focus to optimize outcomes for all lumbar fusion procedures including revision situations.
Previous Minimally Invasive Surgery
In modern medical settings there is an increasing focus upon minimally invasive surgery, which appropriately aims to reduce the trauma of the surgical approach, reduce hospital stay, achieve rapid rehabilitation and optimize outcomes.15 Unfortunately, minimally invasive surgery often involves restricted visualization and may well have a long learning curve. Inadequate exposure can be related to loss of position and wrong level surgery, inadequate decompression, sacrifice of anatomical structures for visualization, incomplete or inaccurate instrumentation, inadequate preparation of posterior fusion beds, and inadequate deformity correction. These possibilities must be considered for all patients, but particularly for the patient who has had a minimally invasive approach where the primary surgeon had not yet achieved mastery of this evolving technology.
PATIENT HISTORY
Overview
As in the presentation of any spine patient, the history is paramount when assessing the Failed Spine Surgery patient. The following plan is a useful formula for organization of the patient narrative. It is useful to have the patient ‘tell their story’ from the onset of their lumbar spine problems through to the present. This narrative will likely involve previous treatments and their efficacy. When there has been previous surgery, it is particularly important to clarify the specifics of each surgery. While the nature and level of the operation is obvious, it is very useful to have the patient detail their specific symptoms (e.g., back- or leg- dominant pain) prior to surgery, and obtain information on if and for how long there was any improvement in the pre-operative complaints. Any complications should be noted for each operative intervention.
Current Symptoms
Have the patient detail their current symptoms and, in particular, have them indicate the site of the symptoms by pointing. Patients often have confusing understandings of medical terms and might, for example, describe ‘hip’ pain when they point to the buttock, and ‘sciatica’ when pain is limited to the buttock only. Establishing the dominant site of pain when there is both back and leg pain is clearly important. Neurological symptoms will assist in diagnosis if there is a suspicion of nerve root compression. A focus on exacerbating factors is also valuable. Back-dominant pain that is worse with bending or sitting may indicate the disc as a source of symptoms, while back-dominant pain worse with standing and walking, but relieved by sitting, may implicate the facet joints. Leg dominant pain worse with standing and walking is recognized as representing neurogenic claudication associated with spinal stenosis. Back-dominant pain associated with standing and walking, and relieved by sitting or the use of a shopping trolley, is often misinterpreted as claudication when it is in fact pain from the posterior elements of the lumbar spine – normally the facets. This latter clinical syndrome is typical in patients who are compensating for sagittal malalignment. Decompression of any stenosis alone in such patients will often make the patient’s clinical syndrome worse.
The history of sphincter function is also mandatory, and any departures from normal must be established. This history alone will give major clues as to any neurological sphincter dysfunction, yet with clarification, the more common symptoms of urge or stress incontinence, prostatism or bladder instability may be clarified.
Treatment – Past and Present: Careful attention to symptomatic treatment with medication and physical interventions is required. Previous attempts at rehabilitation need clarification, as do the patient’s attitude to and compliance with such programs.
General Medical Condition: An overview of health status is necessary. Smoking and other medical comorbidities influence responses to treatment in the past, and potentially in the future. The aged patient with multiple medical comorbidities represents a major challenge in terms of fitness for surgery. Many of the reconstructions required, particularly in the presence of deformity, will be major and require expert medical support.
Employment and Activity Level: For the working age patient a detailed understanding of work history is essential. The level of education, work types, time off work around procedures and time away from employment at presentation will all indicate important patient characteristics and expectations. Worker’s Compensation claims and legal representation are well recognized as being associated with poor return-to-work rates and reduced functional recovery.16,17 Activities of daily living both in and outside the home mirror functional activity. Sport and exercise also indicate past and current levels of function and patient tolerance to effort.
Health Related Quality of Life Outcomes: It is very useful to have baseline scores for all patients so as to map changes and improvements with intervention. There are a wide range of metrics that measure subjective symptoms and function and may be either disease specific or measure general health and function. One typical subjective score is the pain score (scale 0–10), which may be separated into ‘back’ pain and ‘leg’ pain. Disease specific measures of function for the lumbar spine include the Oswestry Disability Index (ODI),18 the Roland Morris Disability Questionnaire (RMDQ),19 the Japanese Orthopaedic Association Score (JOA), and the Low Back Outcome Score. Measures of general health include the SF36, the SF12, The Euroquol, and the COMI score. All of these functional measures attempt to place a numerical score on a level of function. Serial use of such scores in the Failed Spine Surgery patient indicates level of disability and allows the tracking of progress throughout treatment.
Documentation: In the current environment of information sharing, recent documentation of intervention is a useful review of the previous surgeon’s intentions, operations and findings. Documentation of complications is useful, as are records of the patient’s response to perioperative care. Particular attention should be paid to the brand and type of previous instrumentation, as availability of correct instrumentation is vital at the time of any revision surgery.
CLINICAL EXAMINATION
The principles of examination of the patient with a lumbar spine condition are well established, however there are some useful tips for patients with a history of previous surgical intervention.
Overview: Observe the patient’s demeanor during the consultation. Inability to sit for the consultation is a significant marker of disability. Observe for scars from surgery and bone graft donation to confirm that it correlates with the history of previous interventions. Mottled discoloration of the skin over the low back (erythema ab igne) is an indicator of the application of heat – hot water bottles or wheat bags – that have been used for a prolonged period for symptomatic improvement. Leg length assessment and coronal balance may reflect previous lower limb disorders or surgery and scoliosis. A clinical assessment of sagittal alignment is useful; ensure that the patient has fully extended knees and ask them to stand comfortably. It may be that there is minimal stoop, but casual enquiry (often to the partner or spouse) may well reveal that the degree of stoop is significantly greater with fatigue or as the day goes on. The patient will likely initially demonstrate their best sagittal alignment during the examination, but that may not reflect their posture during day-to-day activities. Gait patterns will give clues as to pain, leg length and neurological deficits (foot drop/Trendelenburg). Heel and toe walking are gross measures of anterior compartment and calf muscle strength. More detailed assessment of subtle motor loss can be elicited with repeated single level calf raises, and to assess quadriceps strength, single leg knee bends.
Lumbar Range of Motion: Having the patient flex to bring his or her fingertips towards the floor with the knees fully straightened tests both lumbar and hip flexibility. This is normally a synchronous motion, but significant departures are recognizable – the rigidly maintained lordosis in the presence of excellent hip and hamstring flexibility versus the lumbar spine that unfolds fully in flexion in the presence of limited hip flexion due to tight hamstrings. Specific measures are unlikely to be useful. However, a global measure of fingertip to floor distance or an observation of the reach of the fingertips in relation to regions of the leg is a useful baseline. Extension to the neutral upright posture, from the position of maximum flexion, may reveal insights. Occasionally this is the more painful maneuver and patients may ‘walk’ their hands up the thighs to regain the upright posture. Sometimes termed ‘clinical instability’, this pattern of movement seems to correlate with posterior facet joint pathologies, although the scientific validity and specificity of such findings is clearly limited. Regardless, it is still a very useful patient-specific limitation to observe and document. Extension from the neutral does decrease with age; however, when it is limited relative to the patient’s age and associated with back pain, particularly when combined with alternating lateral rotation from side to side, it is an indicator of potential pain generation from the posterior anatomical structures (normally the facet joints). Conversely, lumbar extension combined with ipsilateral lateral flexion that reproduces or exacerbates leg-dominant pain putatively narrows the lateral recess and foramen and may be clinically indicative of spinal stenosis. Each of these limitations in motion has imperfect scientific validity in terms of diagnostic accuracy, but they do represent repeated patterns in clinical examination and may be useful clues as to the etiology of the pain generator.
Neurological Examination: Baseline documentation of the neurological status in the Failed Spinal Surgical patient is essential. Evaluation of lower limb sensation, motor function and reflexes will document any lower motor neuron loss of function. It is vital to clarify, if possible, whether any documented deficits are recent or long-standing, and their relationship to previous clinical presentations and surgery. Evaluation of upper motor neuron function assessing hyper-reflexia, increased tone, clonus and a positive Babinski response is vital. Most experienced clinicians will have examples of lower limb neurological loss that has defied clear explanation until more proximal pathology has been uncovered with upper motor neuron findings during lower limb neurological examination. Sphincter examination of motor and sensory function along with voluntary motor control is necessary when indicated.
Nerve Root Tension: Limitation to the straight leg raise (SLR) by leg pain will most commonly appear in the recurrent disc prolapse. The cause of the limitation of the SLR must be clear to the examiner. Back pain is frequently limited in the previously operated-upon patient and is not a true positive SLR finding. Hamstring tightness will also be common in the deconditioned patient with prolonged disability. The femoral nerve stretch test (FNST) reproducing anterior thigh pain is often found in mid-lumbar disorders with nerve root compression commonly seen after previous lower lumbar fusion. Conversely, the FNST that reproduces back pain rather than anterior thigh pain again causes extension of the lumbar spine and may further implicate the facet joints that are likely loaded during this maneuver.
Palpation: Palpation of the low back is best performed with the patient prone, the head resting, and the paraspinal muscles relaxed. Localized tenderness may provide helpful information. Hypersensitivity to light touch may reflect overall patient distress. Implant tenderness can be localized and severe. Other sites of tenderness are also worthy of note. Bone graft donor sites may be tender. Lateral hip pain after reconstruction of the lumbar spine may represent a change in hip abductor function, be associated with trochanteric tenderness, and be consistent with ‘trochanteric syndrome.’20
Overview of Other Systems: It is necessary to exclude abdominal, vascular and hip pathologies as a cause of low back and lower limb pain. In the previously operated-upon patient, new or ‘uncovered’ hip arthritis may mimic upper lumbar nerve root compression with anterior thigh pain. Older patients frequently have multiple musculoskeletal problems. While a lumbar problem may appear isolated at initial presentation, successful treatment of the spinal pathology will increase function and activity, often unmasking other musculoskeletal problems such as hip osteoarthrosis.
Abnormal Illness Behavior: Patients may manifest distress through abnormal illness behavior; there have been a number of clinical tests devised to detect this. Failure to achieve expectations with previous surgery will increase the psychosocial stress in many patients and this may be manifested in clinical examination findings. The Waddell tests are useful: axial pressure exacerbating low back pain; pain on whole body rotation; ‘give way’ to manual muscle testing and non-anatomical sensory loss are indicators of abnormal illness responses to clinical examination. Their specificity and sensitivity are unclear, however when combined with other indicators including patient history, exacerbation of symptoms with all physical treatments and widespread pain on the pain diagram, they give an additional indicator to psychosocial distress that may compound the underlying pathology.
IMAGING
When evaluating the Failed Spinal Surgery patient, it is highly likely that multiple imaging modalities will be necessary. The use of several modalities is particularly prevalent when previous fusion surgery has been performed or attempted. Only in the most simple of revision situations – such as the recurrent disc prolapse – will only limited imaging be required. It is helpful to explain to the patient that their clinical problem requires imaging from several perspectives: radiographs to look at spinal alignment and previous instrumentation; MRI to assess neural compression and the status of adjacent unoperated levels; CT scan to look at bone anatomy and fusion status; Nuclear medicine to look for increased bone biological activity perhaps representing pain generators; and possible pain provocation procedures and diagnostic anesthetic injections again to assist with identification of ‘pain generators’. The patient will be helped by understanding that their problem requires examination from several aspects in order to understand previous events and potential sources of current disability so as to optimize the likelihood of success with any revision intervention.
Radiographs
While the role of the X-ray in diagnosis has always been limited in lumbar spine disorders, it plays an extremely important role in assessment and planning, particularly in revision situations. Key advantages include clarification of segmentation and levels; identification of listhesis – anterior spondylolisthesis, retrolisthesis and lateral listhesis with a scoliosis; definition of coronal deformity, including asymmetrical disc space collapse and scoliosis; and assessment of sagittal alignment. Apart from the clarification of lumbosacral segmentation, all other imaging should be performed erect with the patient standing. The pelvis must be included. Sagittal alignment should be assessed with full length spine views, the relationship between the pelvic anatomy and the lumbar lordosis established, and pelvic tilt measured as a metric for compensation for loss of lordosis that occurs through the hip. Whenever revision surgery may require fusion, or previous fusion has been attempted, assessment of the PI/LL/SVA/PT (Figs. 1-1 and 1-2) and their incorporation in surgical planning are necessary. Flexion/extension radiographs have a place for some practitioners as they can determine end range hypermobility, but any role for diagnosing ‘clinical instability’ remains controversial. Flexion/extension radiographs may be useful to assess nonunion, although the lack of measurement precision certainly makes them less helpful than when assessing anterior nonunions in the cervical spine. Assessment of fusion by plain radiographs is of questionable value. Occasionally, very obvious fusion is clear with posterolateral bridging bone and interbody fusion, but since overlying soft tissue and bony shadows often make interpretation difficult, the accuracy of plain radiographs to assess lumbar fusion is debatable. Radiographs are, however, useful to determine deformity correctability. Side bending radiographs can document curve flexibility in scoliosis and kyphosis mobility in the thoracolumbar and thoracic spine. Correlation between erect and supine radiographs (or supine MRI) gives a further indication of spinal flexibility.
Magnetic Resonance Imaging (MRI)
MRI remains the major imaging modality for assessment of neural compression secondary to disc prolapse or spinal stenosis. It is also essential to document the condition of adjacent discs and any degenerative changes. Other useful findings in the Failed Spinal Surgery patient include pseudomeningocele, arachnoiditis, and identification of the dural tube in relation to other structures where previous decompression has been performed. Contrast enhancement is most useful to differentiate between postoperative perineural scar or fibrous tissue versus recurrent disc prolapse. It is also useful to examine the disc space and adjacent vertebra for any suggestion of deep infection. MRI is, however, limited in utility when there has been previous instrumentation. Titanium implants have a less disruptive influence upon imaging than older stainless-steel implants, yet assessment of segments proximal to pedicle screw instrumentation remains challenging.
Computerized Tomography (CT)
While CT scanning may also be used to look at neurocompression, its major utility for the Failed Spinal Surgery patient is to examine the bone anatomy. Bone windows on CT give excellent visualization of the normal osseous anatomy, previous regions of bone resection, and bone landmarks useful at the time of revision surgery. Previous fusions are readily evaluated by CT. CT clearly demonstrates the ‘flowing’ bone mass of a previous posterolateral fusion, bone fusion across a facet joint in an axial image, and continuous trabecular fusion within an interbody space on both sagittal and coronal reformatting sequences. Three dimensional reconstructions improve visualization of bone anatomy in the setting of previous surgery and can be very useful when planning revisions. CT scans are considerably less interfered with by in situ metal (particularly titanium) instrumentation. The CT will give valuable insight into the accuracy of placement of pedicle screws, the location of previous pedicle screw tracts, and the potential for neural impingement. Adjacent segment stenosis may also be usefully imaged with CT, even when MRI has been degraded.
Myelography
The role of water soluble myelography in lumbar spine evaluation has diminished markedly with the widespread availability and improved quality of MRI. However, it does have an infrequent role, particularly if a patient has suspected stenosis adjacent to a fusion with stainless steel instrumentation (Fig. 1-4). Myelography also has the advantage of permitting a degree of axial loading during the investigation due to tilting of the table, and this may uncover subtle neurocompression. It should be combined with CT imaging to optimize cross sectional information. In the presence of a pseudomeningocele, a myelogram may help identify the ‘neck’ of the meningocele.
FIGURE 1-4. CT/Myelogram with sagittal reconstructions to demonstrate spinal stenosis (arrow) at the top end of the instrumentation. MRI will not see this detail because of the interference in image quality due to the stainless-steel implants.
Nuclear Medicine
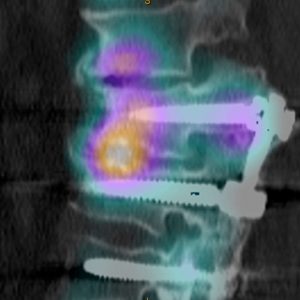
Nuclear medicine bone scans have the potential to demonstrate areas of increased ‘bone activity.’ When combined with CT scanning, the Single Photon Emission CT (SPECT scan) can precisely localize a site of increased bone turnover (Fig. 1-5). While the presence of a ‘hot’ bone scan is not necessarily specific in terms of diagnosis, it provides useful supportive information. After previous interbody fusion with ongoing back-dominant pain and questionable bridging bone on CT, a hot bone scan supports a diagnosis of nonunion. In the aforementioned clinical scenario, but with the addition that the patient’s pain is made worse with prolonged standing and lumbar extension and yet the CT scan strongly suggests a good interbody fusion, increased uptake in the adjacent facet joints implicates the adjacent segment as the likely source of ongoing pain. Similarly, a negative bone scan may be useful to exclude pathologies purported to be responsible for patient symptoms. A further imaging modality that exploits bone biological activity is the Sodium Fluoride CT-PET (Positron Emission Tomography) scan. While the focus for PET scanning is metabolic hyperactivity representing oncological disorders, the use of NaF-18 gives dramatic imaging of bone biological activity that can be very useful in Failed Spinal Surgery Patients.21,22
Pain Provocation Studies
The most common pain provocation study for the Failed Spine Surgery patient is the Discogram. Discography aims to provoke concordant pain reproduction for patients with ‘discogenic’ pain origin by injecting contrast within the center of the lumbar disc under radiological control in the conscious patient. The reliability of this modality has been hotly debated in recent decades, and its less common use correlates with improved quality and accessibility of MRI. It can, however, be useful, and this author makes infrequent use of it to help identify the pain origin, particularly in patients with flexion-aggravated back-dominant pain adjacent to a fusion or to a nonunion. In the latter circumstance, the value of the discogram is in surgical planning. Similarly, a patient who has multiple abnormal discs in the setting of previous failed surgery, and who has multiple positive responses to discography, may well be best advised to avoid any further intervention – the disability associated with extensive revision surgery that includes all positive discs has a low likelihood of successful pain and disability reduction. In such a case, the discogram helps define an absence of precision in diagnosis and indicates the wisdom of avoiding further intervention. The use of a control discogram in an adjacent normal segment is generally avoided due to the evidence that the disc puncture itself is harmful to the structure and future of the normal disc.23
Nerve and Facet Joint Blocks
Using anesthetic blocks to relieve pain from various structures can give useful information as to the likelihood of those structures being responsible for the patient’s symptoms and help in evaluation of the Failed Spine Surgery Patient. Upper lumbar nerve root blocks that successfully relieve pain for a period that correlates with the duration of effect of long acting anesthetic may help clarify if thigh pain is of a spinal or hip origin. Disc space narrowing at the lumbosacral junction is common in asymptomatic individuals, yet may contribute to L5 root pain due to foramen stenosis. If a patient has had previous discectomy at that level without a central neurocompressive lesion, an L5 root block may give further support to the diagnosis of foramen stenosis. A positive response to a facet injection of anesthetic, in association with a hot bone scan and a clinical presentation of back-dominant pain worse with extension, may indicate the facet as a pain source within a previously decompressed level or adjacent to a fusion. The accuracy of these tests is not perfect, and the placebo effect plays such a role that many have suggested the importance of placebo-controlled investigations, although this practice is not currently in widespread clinical use.
Other Musculoskeletal Site Imaging
While there is potential for various additional lower limb investigations that might be used to exclude the spine as the primary source of ongoing pain after previous surgery, evaluation of the hip joint is certainly worth acknowledging. Patients with lateral hip pain often have findings consistent with ‘trochanteric syndrome’, and patients with anterior thigh pain often have hip joint pathology that may masquerade as upper lumbar nerve root pain. Hip radiographs, hip and trochanteric MRI, diagnostic and therapeutic trochanteric injection with anesthetic and steroid, and anesthetic hip arthrogram all have a role when there is a high clinical suspicion of overlap between spine and hip origin to symptoms.
SUMMARY
It is clear that the evaluation of the FSS patient will vary from a relatively straightforward assessment to a highly detailed clinical evaluation and requirement for multiple imaging modalities. For instance, a diagnosis of recurrent disc prolapse requiring repeat microdiscectomy may be fully imaged with only a repeat MRI scan with contrast. At the other end of the spectrum, a patient who has had previous fusion and has a high suspicion of sagittal malalignment will require extensive imaging to adequately identify the ‘pain generator’, determine any malalignment, and optimize understanding of the soft and hard tissue anatomy to fully comprehend the altered anatomy created by previous surgical intervention.
For the surgeon, this process of evaluation can be both interesting and rewarding in terms of precision of diagnosis that enables patient-specific planning of revision surgery. For the patient, the clear demonstration of a highly detailed, thorough, and organized process will give great confidence in a situation of great pain and disability that will likely be further influenced by previous surgical experiences. For both the patient and the surgeon, clarity of diagnosis resulting in precision planning for revision surgery – when indicated – will offer the best outcomes. Accuracy of assessment and subsequent planning are paramount, and there is no role for revision intervention upon the basis of desperation and hope alone.
REFERENCES
- Terran J, Schwab F, Shaffrey CI, et al. The SRS-Schwab adult spinal deformity classification: assessment and clinical correlations based on a prospective operative and nonoperative cohort. Neurosurgery. 2013;73(4):559-568.
- Schwab F, Ungar B, Blondel B, et al. Scoliosis Research Society-Schwab adult spinal deformity classification: a validation study. Spine. 2012;37(12):1077-1082.
- Duval-Beaupère G, Schmidt C, Cosson P. A Barycentremetric study of the sagittal shape of spine and pelvis: the conditions required for an economic standing position.
- Ann Biomed Eng. 1992;20(4):451-462.
- Roussouly P, Gollogly S, Berthonnaud E, Dimnet J. Classification of the normal variation in the sagittal alignment of the human lumbar spine and pelvis in the standing position. Spine. 2005;30(3):346-353.
- Smith JS, Klineberg E, Schwab F, et al. Change in classification grade by the SRS-Schwab Adult Spinal Deformity Classification predicts impact on health-related quality of life measures: prospective analysis of operative and nonoperative treatment. Spine. 2013;38(19):1663-1671.
- Lazennec JY, Ramaré S, Arafati N, et al. Sagittal alignment in lumbosacral fusion: relations between radiological parameters and pain. Eur Spine J. 2000;9(1):47-55.
- Bourghli A, Aunoble S, Reebye O, Le Huec JC. Correlation of clinical outcome and spinopelvic sagittal alignment after surgical treatment of low-grade isthmic spondylolisthesis. Eur Spine J. 2011;20 Suppl 5:663-668.
- Kawakami M, Tamaki T, Ando M, Yamada H, Hashizume H, Yoshida M. Lumbar sagittal balance influences the clinical outcome after decompression and posterolateral spinal fusion for degenerative lumbar spondylolisthesis. Spine. 2002;27(1):59-64.
- Rothenfluh DA, Mueller DA, Rothenfluh E, Min K. Pelvic incidence-lumbar lordosis mismatch predisposes to adjacent segment disease after lumbar spinal fusion. Eur Spine J. 2015;24(6):1251-1258.
- Damasceno L, Catarin S, Campos A, Defino H. Lumbar lordosis: a study of angle values and of vertebral bodies and intervertebral discs role. Acta Ortop Bras. 2006;14(4):193-198.
- Anwar HA, Butler JS, Yarashi T, Rajakulendran K, Molloy S. Segmental Pelvic Correlation (SPeC): a novel approach to understanding sagittal plane spinal alignment. Spine J. 2015;15(12):2518-2523.
- Yang X, Kong Q, Song Y, Liu L, Zeng J, Xing R. The characteristics of spinopelvic sagittal alignment in patients with lumbar disc degenerative diseases. Eur Spine J. 2014;23(3):569-575.
- Roussouly P, Gollogly S, Berthonnaud E, Labelle H, Weidenbaum M. Sagittal alignment of the spine and pelvis in the presence of L5-s1 isthmic lysis and low-grade spondylolisthesis. Spine. 2006;31(21):2484-2490.
- Watkins RG 4th, Hanna R, Chang D, Watkins RG 3rd. Sagittal alignment after lumbar interbody fusion: comparing anterior, lateral, and transforaminal approaches. J Spinal Disord Tech. 2014;27(5):253-256.
- Phillips FM, Cheng I, Rampersaud YR, et al. Breaking through the “Glass Ceiling” of minimally invasive spine surgery. Spine. 2016;41 Suppl 8:S39-43.
- Manson JF, Landham PR, Cunningham JE, Montgomery AS, Don AS, Robertson PA. Universal no-fault compensation is associated with improved return to work rates in spine fusion. Spine. 2015;40(20):1620-1631.
- Montgomery AS, Cunningham JE, Robertson PA. The influence of no fault compensation on functional outcomes after lumbar spine fusion. Spine. 2015;40(14):1140-1147.
- Fairbank JC, Pynsent PB. The Oswestry Disability Index. Spine. 2000;25(22):2940-2952.
- Chapman JR, Norvell DC, Hermsmeyer JT, et al. Evaluating common outcomes for measuring treatment success for chronic low back pain. Spine. 2011;36(21 Suppl):S54-68.
- Redmond JM, Chen AW, Domb BG. Greater trochanteric pain syndrome. J Am Acad Orthop Surg. 2016;24(4):231-240.
- Grant FD, Fahey FH, Packard AB, Davis RT, Alavi A, Treves ST. Skeletal PET with 18F-flouride: applying new technology to an old tracer. J Nucl Med. 2008;49(1):68-78.
- Strobel K, Vali R. (18)F NaF PET/CT versus conventional bone scanning in the assessment of benign bone disease. PET Clin. 2012;7(3):249-261.
- Carragee EJ, Don AS, Hurwitz EL, Cuellar JM, Carrino JA, Herzog R. 2009 ISSLS Prize Winner: Does discography cause accelerated progression of degeneration changes in the lumbar disc: a ten-year matched cohort study. Spine. 2009;34(21):2338-2345.