Sanghita Bhattacharya, Vikas Kaul, Constantine K. Demetropoulos, and Vijay K. Goel
INTRODUCTION
Back pain is a major public health problem in industrialized societies. This places an enormous economic burden on the society. In adults 17 to 64 years of age, back or spine impairment accounts for a greater percentage of absence from work, extended leave and loss of productivity than any other medical ailment.1,2 Almost 30 million people in the United States suffer from low back pain and approximately 10% of these individuals suffer from chronic disabling back pain.1 Back pain has a strong correlation to intervertebral disc degeneration.3,4 Disc degeneration is often associated with disc herniation and sciatica but is often asymptomatic.4,5 In general, spinal disorders reduce mechanical stability, leading to abnormal motion and pain during daily activities. Some spinal disorders may be associated with physical deformity.3 Back pain-related diseases account for an annual cost of approximately $50 billion dollars in the United States alone.6 Degenerative disc disease (DDD) leading to spinal stenosis in elderly patients is a primary reason for age-related disabilities.7
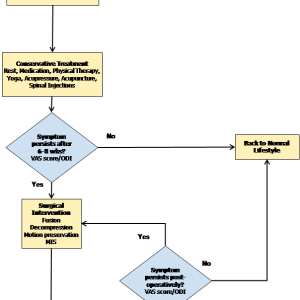
In the early stages of low back pain, rest, physical therapy and anti-inflammatory medications are the first line of conservative, non-surgical treatment. When conservative treatment fails to alleviate pain, surgical intervention may be the only remaining option to regain a functional lifestyle. Non-surgical treatment may prove unsuccessful in the treatment of degenerative disc disease (DDD), particularly when spondylosis or spondylolisthesis is present. An individual suffering from low back pain that affects his daily activities for a prolonged time makes an ideal candidate for surgery. Failure of conservative therapies leads to surgical intervention. Post-surgery follow-up involves evaluation of the relative pain score (VAS – Visual Analog Score) and the functional score (ODI – Oswestry Disability Index), after which the patient might be recommended for a revision surgery. The final aim is to alleviate pain and to restore a normal lifestyle. A simplistic algorithm to elucidate the treatment options has been depicted in Figure 3-1.
Currently, a wide variety of spinal instrumentation is available for treatment of various spinal disorders. The importance of biomechanics and other mechanical factors that contribute to the sustainability and performance of spinal implants cannot be underestimated. Stress, strain and failure analyses of these implants are performed in bench-top, animal, cadaveric or numerical studies to justify the efficacy of these devices in vivo. The range of spinal ailments that are treated using instrumented spinal surgery is vast. These include stenosis, disc herniation, spondylosis, spondylolisthesis, scoliosis or trauma-related injuries including those associated with automotive accidents. The aim of this chapter is to concentrate primarily on DDD (its associated indications such as spinal stenosis and disc herniation) and the biomechanical aspects of devices that have been designed to treat this degenerative condition.
SURGICAL INTERVENTION: DEGENERATIVE DISC DISEASE
A functional spinal unit (FSU) consists of two adjacent vertebrae, the intervertebral disc and the adjoining ligamentous soft tissues. The FSU is thus composed of three separate articulations: two facet joints and an intervertebral disc. The vertebral body acts as the principal load-bearing member. The vertebral canal is surrounded by the posterior aspect of the vertebral body and the posterior elements of the vertebra. This ossified canal protects the spinal cord along its entire length. The normal intervertebral disc is an isotropic structure; the jelly-like nucleus pulposus acts like a fluid-filled bag and swells under pressure. This pressure transmits as a circumferential tension to the annulus, converting it to a load-bearing structure. This whole setting acts as a shock absorber for the spine, such that there is no high spot loading at any point, and allows for complex motion to occur. The intervertebral disc also acts to constrain this motion, adding to the stability of the motion segment (or the FSU). A degenerated annulus can have fissures, microscopic fragmentation of individual fibers. Annular tears at the corners of the vertebral body separating the annulus from the endplates (due to age, wear and tear), concentric cracks, cavities, and radiating ruptures are seen. Subsequently, the annulus bears higher axial compressive loads that lead to high shear forces in the concentric lamellae8,9 and, as a consequence, loss in the mechanical integrity of the disc. Disc bulging may occur due to decrease in the radial tensile strength of the annulus. The degeneration of the nucleus occurs due to loss of water content of the nucleus. Nucleus degeneration in association with the annular degeneration may cause disc herniation which leads to nerve impingement. Thinning of the disc and a loss of disc also occurs in a degenerated disc. Loss of disc height combined with gradual ossification of the endplate and protrusion of the disc tissue causes stenosis, also leading to back pain.10-12 Once degeneration sets in, the intervertebral disc goes through a cascade of degenerative changes resulting in the biomechanical alteration of the load transfer through the disc, causing changes in the mechanical properties and composition of the tissue. This structural disorganization leads to the failure of the hydrostatic mechanism.12
Disc degeneration can eventually lead to reduced disc height, compression of the neural elements and spinal stenosis. Even though the exact pathogenesis of the degenerative process is still unknown, the factors that might cause degeneration are aging, mechanical factors due to occupational exposure, abnormal loading conditions and the loss of nutrition to the disc.10,12 It has been shown that disc degeneration might also be genetically predetermined.13 Disc degeneration and facet joint osteoarthritis are often related, but usually disc degeneration precedes facet joint osteoarthritis.14,15 Biomechanically, the facet joints are important stabilizing structures and carry about 18% of the total compressive load borne by a lumbar spine segment. Facets also are mainly responsible for preventing large extension rotation and shear.16 Higher facet loads and stresses are seen in extension rotation and shear, which might lead to facet osteoarthritis or hypertrophy leading to spinal stenosis. Degeneration of facet joints due to mechanical factors like increased facet loading and wear is called “the facet syndrome.” Spondylosis, spondylolisthesis, disc herniation and spinal stenosis may follow these degenerative changes in the segment.17
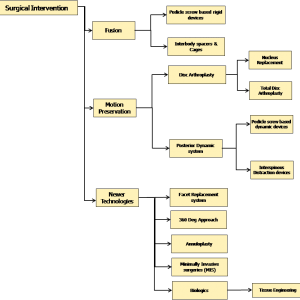
When conservative treatment fails, surgical intervention may be the only remaining option to regain a functional lifestyle. Typically, the goals of such surgeries are to remove the diseased or injured tissue and to restore stability and function to the spinal unit. Spinal fusion is the gold standard of surgical treatment for DDD and is derived from experience at other joints of the body where arthrodesis (fusion) has been used to treat joint pain. In recent years, a variety of internal fixation devices have evolved to achieve improved correction of the deformity, stability and osseointegration at an increased rate. The classification in the field of spinal instrumentation has been summarized in Figure 3-2. This classification provides the entire scope of the chapter.
For decades, fusion (joint arthrodesis) has been one of the most common surgical methods for treatment of degenerated discs. The main clinical problem addressed in spinal fusion is the restoration of stability to a clinically unstable spine. Disc arthroplasty and dynamic stabilization are emerging technologies indicated for younger patients suffering from degenerated disc in more than one segment. They can also be used for patients for whom adjacent level degeneration is a possibility in a long-term follow-up. Current research involves newer technologies such as facet replacements, biologics etc. Each of these surgical interventions is described in detail in the following sections.
FUSION
Evolution of Fusion Instrumentation
As the name suggests, the bony segments of the spine are fused with the goals of deformity correction, pain reduction or elimination, and restoration of stability. Spinal fusion may be accomplished with or without the use of spinal instrumentation. External instrumentation can be used as an alignment and load-sharing device until a bony fusion is achieved.18 This instrumentation has evolved over time and is used in combination with external braces and bone grafts to improve both the rate and degree of bone consolidation.
Harrington, who was the first to utilize hooks and rods to correct spinal deformity and instability, is considered a pioneer of posterior fixation devices for fusion.18 Sublaminar wires were also introduced for fixation procedures.18 However, wires were associated with further complications and did not prove clinically successful. Pedicle screws with rod and plate systems then evolved. These systems were founded on the fact that the pedicles have a strategic advantage in terms of force application in comparison to the facets and laminae.18 (See Fig. 3-2.)
Classification of Fusion Procedures
An effective method of classification for spinal fusion devices is to categorize them according to their point of application, such as anterior, posterior or interbody.19 Another method of classification is based on the surgical approach. Such categories include anterior lumbar interbody fusion (ALIF), transforaminal lumbar interbody fusion (TLIF) and posterior lumbar interbody fusion (PLIF) procedures in the lumbar spine. Posterior fusion techniques include procedures like posterior midline fusions, facet joint fusions, posterolateral fusions and posterior interbody fusions.20 Anterior approach procedures such as anterior interbody fusions are typically employed once posterior surgical procedures have failed and when the motion segments to be fused are kyphotic in nature.20 A combination of an anterior interbody fusion along with posterior fixation was first suggested as a treatment for complicated spinal ailments, but use of this technique has grown to such predominance that it is often used to improve the success rates of fusion procedures in general.20
In a recent comparative work by Wang et al.,21 two groups of patients were treated with either anterior spinal fusion alone or posterior fusion alone. At the two-year follow-up, no significant differences were reported with respect to the clinical efficacy or safety of either procedure. It was suggested that anterior short segmental fusion should be preferred due to reduced surgical trauma as well as reduced implant cost and healthcare expenses. Some investigators have also demonstrated that the posterior approach may also present biomechanical disadvantages, principally due to the concern that the graft material might be susceptible to migration.19 In contrast, posterolateral techniques fuse the transverse processes and facet joints of the operative segment, which avoids the biomechanical concern of graft migration.19
Pedicle Screw-Based Systems
The concept of an optimal load application has led to the development of pedicle screw-based plate and rod systems.18 As these systems do not depend on the laminae or facets, the surgeon maintains the ability to preserve the integrity of the adjacent segments.
Apart from these concerns, surgeons may utilize these systems to apply distraction, compression, shear and bending to achieve surgical correction depending on the clinical scenario.18 In fact, these systems also work in the case of lumbar instability due to both traumatic (e.g., vertebral fracture) and iatrogenic (e.g., spinal tumor resection) causes. Several clinical challenges are associated with these devices. For instance, rigid plates and rods can be difficult to bend and align about all three dimensions intraoperatively, making it a challenge to impart a precise profile to the spine. Misalignment of devices, such as a pedicle screw, can result in serious damage such as spinal canal encroachment.18 Loosening, toggling and wear of pedicle screws have also been documented in several cases.18 Additionally, preventing graft subsidence through the vertebral endplates is an important consideration that can strongly influence the outcomes of spinal fusion procedures.19
While rigid devices are associated with stress shielding and may lead to osteopenia, clinical studies have proven that the rigidity of a device promotes and accelerates the fusion rates. Optimal rigidity can be achieved by providing rigid postoperative fixation while gradually promoting load sharing with time.18 A design derived on the concept of progressive load sharing incorporates the use of polymeric washers placed between the fixation plates and locking nuts. The viscoelastic response of the polymeric washer gradually shifts load to the vertebral bodies while minimizing stress shielding.18 Hardware to restore the complex curvature of the spine remains a challenge. Instrumentation that utilizes shape memory alloys to achieve a complex correction of deformity in multiple axes over time is currently being developed.
Interbody Cages
Apart from pedicle screw-based systems, interbody cages have also become popular as an effective fusion device. Interbody cages are metallic (e.g., titanium) or polymeric (e.g., polyether ether ketone [PEEK]) devices with an empty central region that is packed with bone graft material and then placed in the intervertebral space. These devices have an open design to allow direct bone growth through the cage from one vertebral body into the next.23
A common surgical approach for these devices is the ALIF procedure. Yet, some cages can be inserted using a posterior approach. Traditionally, anterior interbody fusions utilized autologous bone harvested intraoperatively from the patient’s iliac crest. Apart from graft site morbidity, there was a high incidence of nonunion with these procedures. Threaded cylindrical cages designed in the late 1990s improved the success rate of these procedures by providing a more rigid fixation within the disc space. In addition, the amount of bone that was harvested from the iliac crest was reduced, as only the soft inner cancellous bone was needed for the fusion.24
Today, several bone graft substitutes are available, and their use may eventually eliminate the need for bone graft harvests. Threaded lumbar interbody cages, inserted through an anterior approach, were among the first successful interbody cages. Despite years of clinical experience with these devices, it has been demonstrated that threaded cages do not provide ideal fixation in all patients. Outcomes data suggested that the ideal use of these devices was in one-level fusion procedures at L5-S1. Further, L4-L5 fusion was not successful due to a tall disc space and associated risk of isthmic spondylolisthesis.23 In this setting, cages did not provide adequate stability. Moreover, multi-level fusion using these devices required posterior instrumentation. Though initial results involving cages were promising, postoperative assessment demonstrated inferior outcomes associated with the metallic version of these implants and paved the way for alternatives such as PEEK or other radiolucent materials. Yet, these newer devices require additional posterior support.
Cylindrical cages are gradually being replaced by cages with a rectangular geometry, which more closely adapts to the geometry of the disc space. Rectangular geometry with an anatomical contour that conforms to the endplate also increases the surface contact between the cage and the vertebral endplate, resulting in smaller endplate strains. Since cage subsidence is a clinical concern, the reduction of peak strains in the endplates provides a substantive biomechanical advantage.25,26
BIOMECHANICAL EVALUATION OF FUSION DEVICES
In Vitro
Design improvements follow rigorous biomechanical testing and are pursued on an iterative basis that involves design, development and testing. Over time, spinal implants are redesigned using ideas drawn from their in vivo performance. Such changes are meant to overcome flaws in the previous design and to expand the clinical indications of the device. The design aspect includes the consideration of geometrical constraints and the evaluation of device-load-sharing upon implantation. Although biomechanical testing offers several advantages, it is the ability to test the weakest link of a complex device that makes it critical to the overall design evaluation. Testing may focus on the bone-device interface, interconnections between device components, or interactions between the device and living tissue. A majority of these data deal with aspects such as construct stiffness, failure load and fatigue load. Besides analytical or numerical models, parametric analyses may also be completed in conjunction with in vitro biomechanical experiments. Animal models are the final step in design evaluation prior to human trials.19 While animal research is associated with ethical concerns, high costs and may present significant differences from studies on human subjects, as such in vivo work may provide data that is otherwise unattainable prior to human implantation. A well-designed animal study will select the appropriate animal model for addressing the testing hypothesis.27 A broad analogy with in vivo human response (i.e., beyond the scope over which the animal model has been validated) should be avoided, and published animal studies should be scrutinized for such error prior to the application of findings.
Axial pull-out studies are commonly used to measure the fixation strength of various screw designs. The pull-out strength of pedicle screws has been extensively studied using cadaveric bone and artificial materials (e.g., polyurethane foam). This serves as a standard procedure to compare the pull-out strength among different designs. Data from these studies have been utilized to improve screw placement techniques, thread designs and a variety of other design considerations. For instance, pull-out tests have been used to develop techniques that use bone cement to improve screw interface fixation. Such techniques have proven useful in patients with poor bone quality.8 For instance, Seller et al.28 affirmed that the outer diameter and insertion depth of the screw should be large enough to attain proper fixation. Other examples of similar work include screw pull-out strength studies conducted by Spivak et al.,29 in which the effects of post-operative fixations were demonstrated in an animal model. Similarly, Yerby et al.30 demonstrated that offset hook usage acts to supplement screw fixation strength. Studies have demonstrated that a close relationship exists between pull-out strength and maximum insertion torque.31 Pull-out studies have also been conducted on other devices. For instance, fusion cage pull-out studies have been used to understand cage migration and expulsion of these devices. Indeed, Brantigan et al. found that a force of 122 N was required to extract a PLIF bone plug compared to 342 N for a carbon fiber cage.32
Testing of osteoligamentous spinal segments provides a means by which the ability of a device to stabilize an injured spinal segment may be quantified. In a typical example of such a study, the quantity of motion (range of motion [ROM]) and quality of motion (neutral zone [NZ], instantaneous axis of rotation [IAR]), intradiscal pressure, facet joint loading and load sharing are measured in the intact spinal segment first and subsequently in the injured and stabilized conditions. Most of these studies are performed under load control. Specifically, pure moments are applied at the top of the spinal construct and the resulting motion is measured. For example, in a comparative study conducted by Schleicher et al., two analogous lumbar interbody cages were compared using a cadaveric model.33 L4-L5 motion segments of 16 human cadaveric lumbar spines were isolated and divided into two test groups. Each specimen was first tested intact, followed by discectomy, and then each device was surgically implanted. The objective of this study was to compare the stability of the standalone SynFix-LR™ ALIF spacer with the previously approved STALIF™ spacer.33 Data from the study suggest that the plate and locking screw configuration of the SynFix-LR™ improves the stability of this implant compared with the STALIF™ under certain loading directions. The stiffness response of the SynFix-LR™ proved to better stabilize the spine in lateral bending, axial rotation and extension than did the STALIF™ device.
Other studies may focus on fatigue loading characteristics or provide data related to failure loading. Thus, Wittenberg et al. subjected five different spinal devices to cyclic flexion-compression loading following an L3 corpectomy in lumbosacral calf spines.34 The AO Schanz, Kluger and Stefee plates were found to be stiffer compared to the Harrington rods or Luque plates. Failure analysis (cyclic flexion and compression loads) demonstrated that four out of the five Kluger devices tested failed in 47,800 cycles or less, and the AO screws failed in 20,800 cycles or less in three out of six tests. No failures were recorded for either Harrington or Luque implants. Nonetheless, the authors asserted that long implants might give rise to strain at the adjacent levels causing early degeneration and destabilization, and therefore, short segment fixation should be preferred in spite of its inherent shortcomings.
Consistent throughout all of these studies is the underlying assertion that pedicle screw systems are only effective over a defined life, irrespective of design variables such as screw size or implant geometry. The implication is that these screws act as fusion devices. Although the device may not be ultimately removed, the post-fusion spine will sustain continued loading. One of the important factors in determining the fatigue life of these systems is the material stiffness.
In Silico
Finite element (FE) analysis is a valuable tool that allows further insight into in situ biomechanics. Models that take into account the geometric and material properties of and contact definitions among the various components of the spinal segment of interest are able to determine a variety of factors including applied loads, implant and tissue stress distribution, joint contact forces, ROM, instantaneous helical axis (IHA) and disc pressure. These studies supplement the knowledge gathered from in vitro experimental studies and facilitate parametric analyses.19 Compared to physical experiments, numerical models offer the ease to readily perform parametric or sensitivity analysis. FE models of intact osteoligamentous motion have been developed by a number of investigators (Goel et al., 35-37 Rohlmann et al.,38 Shirazi-Adl et al.39 and Polikeit et al.40) and subsequently adapted to simulate post-surgical conditions as well.35,41
For instance, Puttlitz et al.19 utilized an L3-L4 FE motion segment to simulate surgical fusion using a range of existing devices. Stress distribution was calculated in the device and the surrounding tissue. Bilateral fusion using unilateral and bilateral plating system was also simulated in FE models. Unilateral models have demonstrated higher peak trabecular stresses than bilateral systems. Similarly, Grosland et al.42 performed a comparative finite element study of multiple interbody fusion cages. The implantation of five different cage designs in a functional spinal unit was modeled, and these models were subjected to compression, flexion and extension. These results demonstrate that various cage designs have resulted in less than significant variations in construct stiffness.
FE technique has also been utilized to understand bone remodeling phenomenon. For instance, adaptive bone remodeling techniques were also implemented by Grosland et al.36 to predict post-operative changes in bone density. It was asserted by the authors that design changes in the implant will have an effect on the loading environment and eventually the bone deposition/absorption rate.
Similarly, a work by Polikeit et al.40 stressed that density of the bone is more crucial in determining biomechanical performance of a cage than the materials used or the spinal loading conditions. FE analysis has been utilized as a tool to determine stress distribution on devices as well as biological tissue. For instance, Zhang et al.43 performed FE analysis to demonstrate the effects of screw parameters on the pull-out strength. Results demonstrated that the major diameter of the screw was much more critical than the pitch and the minor diameter in determining pull-out characteristics. Bench-top tests can also be simulated using the FE method to assist the designer with the development of the device. While FE analysis provides an economical tool that can be rapidly implemented, experimental models are indispensable and provide critical data required for FE validation.
Animal Studies
Animal studies can be used to determine the sequential response of the host tissue following implantation. Therefore, in vivo data can be obtained by histological analysis of the tissue at specified time points. McAfee et al.44 studied the effects of instrumentation on fusion. An in vivo canine model was utilized for this study in which 63 adult beagles were tested with or without spinal instrumentation. The study concluded that instrumented fusion achieved greater rigidity compared to uninstrumented fusion. It was the first study of its kind to prove the occurrence of stress shielding in the spine secondary to spinal instrumentation.44 Issues like biocompatibility can also be analyzed using animal models. For example, Hojo et al.45 investigated the clinical efficacy of a novel bioresorbable cage compared to a carbon fiber cage. During this study, 25 mature sheep and three types of fusion cages were tested. The cages were the autologous iliac bone (AIB), carbon fiber cage (CFC) and the F-u-HA/PLLA (bioresorbable). Histological analysis demonstrated that fusion quality of the bioresorbable cage was equivalent to the other two. This conclusion was formed on the basis of radiographic and biomechanical assessment. Still, the biocompatibility of F-u-HA/PLLA was superior to the other devices tested. It was found that the F-u-HA/PLLA cages had direct contact with the surrounding bone while the CFC was encapsulated in thick fibrous tissue.45
One major limitation of animal studies for orthopedic work is the fact that animal models are typically based on quadrupeds. Clearly a bipedal loading scenario is difficult to evaluate in such a model. Gurr et al.46 simulated corpectomy in an animal model such that the device spanned the entire vertebral space. The surgery was performed in such a way that the device transmitted 100% of the load between segments contrary to the load sharing ability for which it was designed. The conclusion derived from this study was related to the restoration of stability, rather than rigidity after cyclic loading. Nevertheless, the strength of such models is in their ability to assess the effects of altered load conditions on the biological tissues over time. As such, findings should be interpreted for aspects of the experiment that are valid for the selected experimental model.
Motion Preservation
Although fusion was first performed over 100 years ago and still remains the gold standard of treatment for DDD, accelerated adjacent level degeneration, morbidities after spinal fusion, and increased cost due to long postoperative rehabilitation remain a concern.47 Disc arthroplasty is designed to address these concerns. While fusion has shown success in rendering temporary pain relief, it has also shown drawbacks such as pain at the bone harvesting site and adjacent level degeneration. After initial pain relief, pain is likely to return due to spinal stenosis, facet hypertrophy, osteophyte formation, posterior muscular debilitation as well as disc degeneration at the adjacent levels to the fusion site.48-50 Many long-term follow-up studies after the fusion procedure have reported that adjacent level degeneration is a major concern. It is not yet clear if adjacent segment degeneration is due to the iatrogenic production of a rigid motion segment or if it is the progression of the natural history of the underlying degenerative disease.49 Hence, many non-fusion techniques have been investigated and have emerged in recent times to replace the conventional fusion techniques.
EVOLUTION OF DISC ARTHROPLASTY
The traditional belief that a rigid fusion correlates with successful clinical outcomes has recently come under question primarily because of the results drawn from several randomized trials. Limited clinical success rates for spinal fusion and the success of total joint replacements at other joints in the body have motivated efforts to develop joint reconstruction techniques in the spine.51-53 Proponents believe that such techniques will relieve pain and preserve physiological motion. The aim of this process is restoration of disc height, avoidance of accelerated adjacent segmental degeneration and faster recovery.51-53 Disc arthroplasty is an emerging technology indicated for younger patients suffering from degenerated disc in more than one segment. It can also be used for patients for whom adjacent level degeneration is a possibility in a long-term follow-up. As the name suggests, the goal of disc arthroplasty is to completely replace the degenerated intervertebral disc by an artificial implant which has the capability to not only treat the pain-causing symptoms, but also to potentially restore the lumbar motion, unlike fusion.
The earliest clinical literature on disc arthroplasty was reported by Fernstrom in 1966, using a stainless steel ball bearing manufactured by SKF.6 Since then, there has been an ongoing quest for an artificial disc that will not compromise patient safety, will maintain the intervertebral height, will provide an acceptable ROM, will demonstrate long term durability and stability and will restore the energy absorptive qualities of the native disc. The ultimate goal of such a device is to restore function with minimal detriment to surrounding tissue. At the very least, a deep and current understanding of the clinical issues, engineering mechanics and material science are crucial to the mechanical design of a disc prosthetic.53,54
Key factors that must be considered in the design of a disc implant are:
- Each spinal level contains three separate joints – two facet joints and the intervertebral disc;
- The disc in its native state provides a combination of mobility and stability;
- Native disc’s shock absorption capabilities must be mirrored; and
- The IAR is constantly changing as the position of motion segment changes.
CLASSIFICATION OF DISC ARTHROPLASTY
Nucleus Replacements
In nucleus replacement, only the inner portion of the intervertebral disc is replaced by an artificial material. Four different approaches exist for nucleus replacement. In the first technique, the disc is replaced by an inflatable, impermeable bladder filled with fluid. The second approach uses a solid ball-like structure. The third technique uses a partially hydrated, hydrophilic structure which eventually hydrates following implantation and swells to fill the disc space. The fourth and final approach involves the injection of a biomaterial that will eventually polymerize and take on the shape of the disc. Some of the adverse effects associated with these devices are migration of the device due to excessive lateral bulge, small contact area at the bone-device interface that may lead to abnormal stress concentration and instability of the device following implantation.55,56
Some notable devices are described below:
- Raymedica’s PDN™ is one such device that consists of a hydrogel core encapsulated onto a woven polyethylene jacket. A few follow-up studies55 in 2001 confirmed an increase in disc height. While there were initial difficulties related to device migration, these incidents were mitigated by using revised surgical techniques.
- Newcleus™ is a similar device to the PDN™. This device is a polycarbonate urethane spiral coil that is inserted using a posterolateral annulotomy approach after discectomy.
- Aquarelle™ (Stryker Spine, Stryker Corp.), Neudisc™ (Replication Medical, Inc.), Biodisc™ (Cryolife, Inc.) and injectable disc nucleus (Spine Wave, Inc.) are all hydrogel-based devices that hydrate and imbibe water once deployed, taking on the shape and form of the disc space.55
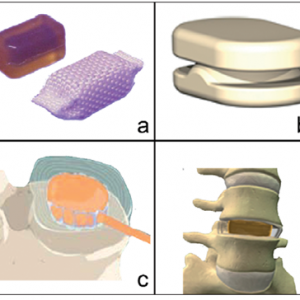
- Prosthetic Intervertebral Nucleus (PIN)™ (Raymedica, Inc.) is an injectable polyurethane formulation that cures after injection. Similarly, DASCOR™ (Disc Dynamics, Inc.) is also a curable polymer that is injected using a balloon placed in the disc space55,56 as shown in Figure 3-3.
Nucleus replacement is a relatively new technology. Although initial results are encouraging, this technique remains investigational. These devices have an advantage in the ease of surgery as compared to fusion or total disc replacement surgeries.
Such a device might serve as an effective treatment for conditions that require more aggressive treatment than a typical herniation, yet do not require disc replacement. Some nucleus prostheses demonstrated problems with migration, extrusion and subsidence, primarily due to the small contact area at the bone-device interface. There have also been cases of excessive lateral bulge and migration of the device. Migration might be due to the loose-fitting nature of the device. As a solution, some devices have developed surface contours to increase surface contact area and congruity.55
Total Disc Arthroplasty
Various disc designs exist on the market. These devices can be categorized on two major design principles. The first group aims to replace the motion characteristic of the disc and is designed with a metal-on-polymer bearing. The second group aims to mimic the viscoelastic response of the nucleus surrounded by the annulus. These devices are designed using polymers.
Constraint is also important and may provide a better scheme for the classification of ball and socket devices (articulating discs) such as Prodisc-L™, Maverick™ etc. Such a classification can be broken down into constrained, semi-constrained and unconstrained designs based on the degrees of freedom.55 The constrained design has a fixed COR (center of rotation). Moreover, the surfaces completely conform and have a retentive interface. The sliding and rolling motion of this artificial joint, in turn, defines spinal motion (see Fig. 3-3)56
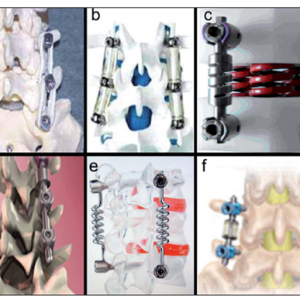
An unconstrained device has a non-conforming ball and socket interface and has a floating COR. The semi-constrained design falls somewhere in between the other two cases57 and allows translational motion in an anterior-posterior direction. Another kind of device is the elastomeric disc such as the Acroflex™ disc. This device was designed with a unique goal of mimicking the shock absorption capacity of a natural disc. A number of disc replacement devices are summarized in Table 3-1 and Figures 3-4 and 3-5.
| Device | Material Couple | Features | Manufacturer |
| LUMBAR DISCS | |||
| SB CharitéTM | Metal-Polymer-Metal | Unconstrained polymeric core and endplates bioactively coated | Depuy Spine, Raynham, MA, USA |
| Prodisc-LTM | Metal-Polymer-Metal | Lordotic angle of the endplates is 6°/11°. Semi-constrained device | Synthes, West Chester, PA, USA |
| MaverickTM | Metal-Metal | COR of this device is fixed. ROM determined by radius of the ball | Medtronic, Minneapolis, MN, USA |
| FlexicoreTM | Metal-Metal | The endplates are plasma coated | Stryker, Kalamazoo, MA, USA |
| MobidiscTM | Metal-Metal | Sliding core and emulates the instantaneous COR (center of rotation) | LDR medical, Troyes, France |
| Activ-LTM | Metal-Polymer-Metal | Sliding core and endplates have spikes or keels | Aesculap AG, Tuttlingen, Germany |
| KineflexTM | Metal-Metal | Semi-constrained disc with a flexible COR | Spinal Motion, South Africa |
| AcroflexTM | Rubber core with titanium endplates | Elastomeric disc | Depuy Spine, Raynham, MA, USA |
| CERVICAL DISCS | |||
| M6TM | Metal-Polymer-Metal | Polymeric fibers emulate the annulus | Spinal Kinetics, Redwood City, CA, USA |
| BryanTM | Metal-Polymer-Metal | Core encapsulated in a polymeric sheath and lubricated with saline | Medtronic, Minneapolis, MN, USA |
| Prestige STTM | Metal-Metal | Two-piece ball-and-trough configuration | Medtronic, Minneapolis, MN, USA |
| Prodisc-CTM | Metal-Polymer-Metal | Keels on the endplates are unique | Synthes, West Chester, PA, USA |
| PCMTM (Porous coated motion) | Metal-Metal | Ridges on the device endplate. Device endplates rectangular in shape and greater in width than in depth emulating the vertebral endplates. | NuVasive, San Diego, CA, USA |
| CervicoreTM | Metal-Metal | Innovative saddle shaped articulation, COR below in flex/ext; above in bending | Stryker, Kalamazoo, MA, USA |
| CervidiscTM (DISCOCERV) | Ceramic-Ceramic | Lordotic 4° angle; endplates have teeth for fixation with hydroxyapatite coating | Scient’X, Maitland, FL, USA |


BIOMECHANICAL TESTING OF DISC ARTHROPLASTY
In Vitro
The quality and quantity of motion following total disc replacement may affect the facet joints of the motion segment and the bone-device interface. Biomechanical evaluation enables us to determine the kinematics of a motion segment as well as the adjacent segments post-total disc arthroplasty (TDA). In a study conducted by Demetropoulos et al.,58 the kinematic properties of the lumbar spine were characterized following Prodisc-L™ implantation. Ten cadaveric spines (L3-L5) were tested under pure moments over a range of ±10 Nm with an applied follower load of 200 N. Load deformation curves as well as the ROM were computed in flexion-extension, lateral bending and axial rotation. This study demonstrated that Prodisc-L™ (semi-constrained) restored flexion-extension motion compared to the intact condition. It was also found that disc insertion led to an increased lordotic angle at the implanted level. Thus, due to increased lordosis, extension ROM was reduced while the total flexion-extension ROM remained unchanged. Although increased lordosis may cause increased facet loading, the restored disc height following surgical disc replacement may compensate for this increase. While lateral bending was not significantly altered, ROM in axial rotation increased slightly. The implantation procedure did not lead to an increased motion or increased disc pressure at the adjacent levels.
In a similar study conducted by Hitchon et al.59 on the Maverick™ artificial disc (constrained disc design), ROM increased in all loading modes. Despite the increase, statistical significance was not established. This study demonstrated that the Maverick™ disc restores spinal motion following discectomy to levels similar to those of the intact state.59 Cunningham et al.60 showed 44% increase in the ROM in axial rotation using the Charité™ disc (unconstrained/floating COR). A marginal increase in all other loading modes was also observed. In this study, the Charité™ disc was compared to the BAK cage™ and the BAK™ cage+ISOLA™ screw rod fixation. The authors stated that the Charité™ disc restored motion to that of the intact segment in flexion-extension and lateral bending. Increased axial rotation was thought to be due to the anterior annular resection that was necessary to implant the device. This study also showed that TDA preserved the kinematics at the operative and adjacent levels compared to lumbar interbody fusion devices.
The post-operative intradiscal pressure (IDP) and ROM at the adjacent levels after TDA is also a topic of clinical importance. For example, in a study on a semi-constrained Activ-L™ device, Ha et al.61 estimated the kinematic response of human cadaveric spines post-implantation. Five L2-S2 spinal segments were tested about all three primary axes of loading. Motion, disc pressure and facet strains were calculated to 8 Nm with a constant 400 N compressive preload. (See Table 3-1 and Figs. 3-4 and 3-5.)
It was shown that, after disc replacement, ROM increased during flexion and lateral bending at the operative level and also at the inferior segment during flexion. Disc pressure at the implanted level was comparable with the intact spine. On the other hand, a trend towards decreased pressure at the inferior disc was observed under all modes of loading but was not found to be statistically significant. Facet strains significantly decreased at the operative level during flexion and axial rotation at the inferior facets. The authors asserted that Activ-L™ arthroplasty was unable to fully restore normal ROM to the intact levels.61
Our prior knowledge of wear-related issues in hip and knee arthroplasty suggests that wear debris, especially polymeric debris, may initiate an inflammatory response leading to periprosthetic osteolysis and bone resorption at the bone-implant interface. Cyclic loading and relative motion at the bearing surface of the device may pose heightened risks due to the proximity of the disc to major blood vessels and neural tissue. Therefore, biotribological tests serve as an effective pre-clinical tool to investigate device-wear characteristics.
Although previously lacking, data on polymeric wear debris has begun to exist for spinal devices. A wear rate of 1.1 mg/million cycles62 has been reported for the Charité™ artificial disc, while the experimental wear rate for the Bryan™ disc was 1.2 mg/million cycles.63,64 For the metal-on-metal Maverick™ disc (constrained), Paré et al.65 reported a steady-state wear rate of 0.33 ± 0.12 mm3/million cycles in flexion-extension and 0.43 ± 0.06 mm3/million cycles in combined motion tests. Though TDA represents a new paradigm in treating disc ailments as we move from an era of arthrodesis to joint preservation, it also presents new challenges such as amount of osseointegration and interface wear rate.
In Silico
Previous work has emphasized the importance of artificial disc designs that preserve the posterior elements. This is of great importance as posterior element degeneration, or the facet joint degeneration, is a common source of low back pain. On the other hand, some studies have reported that apart from preoperative degeneration, the posterior elements may be abnormally stressed by disc replacement. In a computational study performed by Dooris et al.,66 the effects of facet load sharing following TDA were examined. A ball-on-socket disc design by Medtronic was utilized as the implant. Comparisons were made between the pre- and post-implantation FE models. Different annular window sizes and varied anteroposterior artificial disc placements were simulated. Findings demonstrated that an artificial disc can alter spinal bending stiffness in the sagittal plane. Changes in spinal stiffness were noted to be dependent on the position of the disc and degree of annular resection. Anterior placement of the device led to increased facet joint loads in compression and extension. These findings suggest that if the anterior longitudinal ligament is preserved and the implant placed posteriorly within the disc, then the spinal stiffness will be restored and facet loads will be maintained at pre-implantation levels. FE models are an important tool for the computation of complex motions and contact forces in cases where direct experimental measurements are not feasible.
In another numerical study by Denozière et al.,67 a 3D FE model of a two-level osteoligamentous lumbar segment was built to compare cases of arthrodesis vs. TDA using a metal-on-metal ball and socket design. It was found that superior adjacent segment mobility following fusion was reduced by 44% in rotation about all three axes. In contrast, superior adjacent segment mobility increased in all degrees of freedom by 52% following TDA. Interestingly, it was noted that TDA led to high ligament strain, high facet stress and heightened risk of instability. The authors discussed these findings in light of concerns that degeneration associated with these changes in loading following TDA may favor the choice of fusion procedures until more is known. It was also suggested that the COR of an artificial disc should be located posterior to the geometric center of the endplates and that the preservation of the annulus is an important consideration in TDA.67 In a later work by Goel et al.,68 impact of a Charité™ disc replacement at L5-S1 was investigated using the hybrid protocol. (The hybrid protocol involves first testing the intact spine to a given load level, and then repeating these tests for subsequent test conditions such as TDR with a fixed endpoint of displacement determined by the range of motion of the intact specimen.) The FE model predicted an increase in ROM by about 44% at the implanted level and a decrease in facet loads by 13.4%. ROM and facet loads also decreased at adjacent segments.
The impact of the placement of a device on the final outcome is also an important matter of debate. Based on this idea, Le Huec et al.69 utilized an FE model of the lumbar spine to study Maverick™ lumbar total disc replacement. ROM and facet strains were computed. It was proposed by the authors that “less-than-ideal positioning of the prosthesis, especially with an anterior offset, significantly affects the range of motion of the spine segment and causes an increase of inner load in the facets.”
FE models have also been utilized to understand wear characteristics of joint replacements in the hip and knee. Investigators such as Rawlinson et al.,70 Goreham-Voss et al.71 and Bhattacharya et al.72 have used such techniques to understand the in situ wear characteristics of disc implants. FE wear predicting models utilize Archard’s law to simulate steady state wear derived from initial experimental wear rates. The strength of these models lies in the fact that they are a time efficient tool, and, more importantly, they allow the investigator to extrapolate experimental data to much longer wear durations than would be otherwise feasible. Such numerical studies can be utilized to run parametric test cases in order to understand the effects of micro-separation, surgical variability and anatomic variations on a given device’s wear, which is not feasible in mechanical simulators. Despite these advantages, previous FE studies do have their own limitations. For instance, FE models, typically, do not account for muscle forces. Also, patient-specific material properties and anatomical variations in specimens, such as facet tropism, are lacking.
Animal Models
Due to anatomical and postural differences, animal studies are met with criticism though they remain a useful tool for histomorphometric analysis and the investigation of phenomena that must be studied in situ. Non-human primates, such as baboons and monkeys, are probably the best available model, but ethical and economical concerns limit the use of these models.
Animal models are often used to investigate the biomechanical, histochemical and biologic ingrowth characteristics of devices. A review of the literature reveals very few studies on TDA that have utilized animal models. Apart from human cadavers, sheep, baboon, calf and canine spines have been used for such studies. In a study conducted by Taylor et al.,73 thirty-seven adult dogs were divided into three separate groups: intact, fusion and TDA. Animals were sacrificed either immediately after surgery or three months post-operatively. The fusion group demonstrated progressive fusion while the TDA group maintained motion throughout the course of the study. Radiographic and histological analyses demonstrated stable incorporation of the TDA and fusion implants. Yet neither device was fully incorporated. Although the authors assert that full integration could have been obtained six or nine months following surgery, it is difficult to comment on the osseointegration of the artificial discs in comparison to fusion devices.
Non-primate models such as baboons have also been frequently used to understand the degree of porous ingrowth at the device interface. McAfee et al.74 performed TDA (Charité™) on seven mature baboons at the L5-L6 level. This study was designed to determine whether a plasma sprayed hydroxyapatite (HA) or electrochemical HA contoured surface would improve osseointegration of the titanium end plates of the disc replacement. Histopathology after six months demonstrated excellent ingrowth at the bone implant interface for both groups. The assays showed no accumulation of wear debris or cytokines, and the total ingrowth range at the endplate area was 35.5–58.8%. The investigation also revealed that porous ingrowth was more favorable following TDR than following cement-less joint replacements in the appendicular skeleton.74
Similarly, Cunningham et al.75 employed a baboon model to demonstrate the clinical efficacy of the Acroflex™ device. The clinical outcome after 12 months post-implantation showed that this unconstrained device preserved axial rotation, while significantly reducing motion in all other modes of loading. Histomorphometric data proved favorable for this device. The authors claimed that the baboon model represents a worst-case scenario compared to humans because these animals are free to ambulate and perform far more demanding physical activities (e.g., gymnastics typical of a baboon) just one week following the procedure.75
Owing to a paucity of spinal implant retrievals, animal studies serve as a valuable reference for the evaluation of the immunological response to disc replacements. This is an important consideration in the interpretation of implant wear characteristics. Cunningham76 assessed the impact of wear debris on rabbits; a 4 mg bolus of wear debris (300–600 million particles with a diameter range of 0.3–50 µm) was injected into the epidural space. Upon sacrifice after three to six months, no deleterious effects were observed in the surrounding tissue. Several investigators have also suggested that the reactivity of human tissues to such particulates is significantly less than that of rabbits, suggesting possibly higher tolerance limits in humans.62,76 For instance, a 4 mg bolus in rabbits may be equivalent to 50 mg in a 70 Kg human subject.
Many of the abovementioned devices are still undergoing clinical trials, and some have published preliminary clinical data. In some cases, TDA implantations have been reported in which the geometric center of the device and the spine are not aligned properly. Increased segmental lordosis, increased facet loads, subsidence, migration and accelerated articular surface wear have been reported by many groups.77,78 Currently, no significant difference has been reported between constrained and unconstrained designs especially as they relate to the restoration of native biomechanics following implantation. Some studies of cervical TDA have demonstrated altered motion characteristics such as increased ROM at the level of implantation and heterotrophic ossification.78,79 In the case of lumbar TDA, increased facet loads have been documented and semi-constrained devices appear to demonstrate improved load sharing characteristics. However, semi-constrained devices appear to be much more susceptible to wear.78
There is a lot of scope in terms of design and material properties to come up with a novel device that would restore biomechanics of the implanted segment to intact levels and lower the risk of adjacent segment degeneration. An ideal device is expected to redistribute the loads on all the anatomic structures without overloading any of them; load sharing is going to determine the long-term effectiveness of the device. The IAR and the facet loads after disc replacement should be comparable to those before TDR. The intradiscal pressure at the adjacent levels needs to be computed to examine the possibility of adjacent level degeneration post TDA. Nevertheless, based on available studies on various existing designs, it cannot be concluded that total disc replacement is superior to spinal fusion in terms of clinical outcome.
DYNAMIC STABILIZATION SYSTEMS
Evolution of Dynamic Stabilization Systems
As an alternative to TDA, dynamic stabilization systems have begun to gain in popularity. These devices were originally conceived to not only unload the disc at the treated level and reduce discogenic pain but also to restore the IAR. These devices alter the load transmission across the degenerated disc without excision of the disc. They are drawn on the hypothesis that restriction of motion within a safe range will prevent excessive loading and further degeneration.80,81
These devices can be broadly categorized into two groups: pedicle screw-based systems and interspinous spacers. Pedicle screw-based systems alter the entire range motion of the operative segment.55,56 Various devices currently being marketed are unique designs and an understanding of their underlying mechanisms must be considered individually. In contrast, interspinous spacers unload the disc and modify the motion patterns in extension using a physical end-stop. Although there are exceptions, most spacers typically do not constrain flexion.
Classification of Dynamic Stabilization Systems
Pedicle screw-based dynamic system
The Graf Ligament™ system was one of the first dynamic stabilization devices and consists of a non-elastic band used to link pedicle screws between segments such that motion is restricted in full extension. Clinical success of this device was debated primarily because it was unclear whether benefits were due to the transfer of load from anterior to posterior or due to the restriction of motion.55
The Dynesys™ system (Fig. 3-6) is derived from the same concept with minor modifications.55 This device consists of a non-elastic ligament enclosed within a polymeric cylinder connecting pedicle screw heads across levels. The current design consists of titanium pedicle screws, polyester cords (ligaments) and polycarbonate urethane (PCU) spacers. The cords/ligaments act to restrict flexion, while the spacers act to limit extension, thus enabling the posterior elements to be realigned by the device. Biomechanical studies have demonstrated the instrumented segment to maintain the stiffness of the intact spine.82 Though implantations have proven safe and encouraging, screw loosening as well as loss of lordotic angle due to distraction from the spacers is also a concern.
The fulcrum assisted soft stabilization (FASS™)55 was developed as an improvement over previous designs by utilizing a fulcrum between the pedicle screws in front of the ligament. The fulcrum serves to distract the posterior annulus, and the FASS™ system unloads the disc without requiring the assistance of muscle loading. It must also be noted that greater unloading of the disc with the help of an elastic ligament may increase stiffness of the motion segment, thus resulting in screw loosening.55 For this reason, partial unloading of the disc and restoration of mobility to the segment might be a preferable option. This device is still under further development and clinical investigation.
Additional semi-rigid metallic devices are presently under development. One such device is the dynamic soft stabilization (DSS™) system with two design generations, namely DSS-I and DSS-II. The first-generation system has a C-shaped titanium spring, while the second has an elliptical-shaped coiled spring between the pedicle screws; spring flexibility limits the ROM of the segment. The underlying concept here is to match the implant COR with that of the segment in an effort to promote disc unloading and a uniform ROM. Additionally this device is such that a minimally invasive practice is required for its implantation, reducing morbidity associated with the procedure.55
The Isobar TTL™ shown in Figure 3-6 is another notable device in this category, comprised of a titanium alloy rod and a dampener element.55 The dampener element is formed of a series of helical springs that allow linear and angular motion and serve as a shock absorber. This instrumentation allows flexion-extension and axial rotation while lateral bending is restricted. A lordotic angle is also incorporated into this system. Benefits associated with this device include ease of implantation, motion segment stabilization, maintenance of lordotic angle, load sharing and conformance to the IAR of the motion segment.55 Other notable devices in this category include the BioFlex™, Cosmic™ and Stabilimax™ systems.
Interspinous Distraction Device
The first interspinous device was designed in 1986 in Europe, and it was comprised of a titanium block pushed between the spinous processes. Following preliminary success, the material was changed from titanium to PEEK due to its more desirable mechanical properties. The device was secured using a polyester band wrapped around the processes. This new interspinous device was called the Wallis™ device (Fig. 3-7).83 These devices have been categorized as floating devices84 as they are not connected to the vertebrae by a rigid mechanism. They function by distraction of the spinous process, thus unloading the disc at that level. Moreover, the possibility of loosening within these devices has been eliminated.
This design was further modified to incorporate grooves and notches at the edges of the PEEK block in order to achieve a more congruent anatomical fit with the spinous process. The flat bands from the first design were replaced by woven bands with a new system of attachment for the bands onto the spacer block. No torque was generated in the new attachment process due to the tightening process at this interface. PEEK is not only biocompatible but also radiolucent. This makes PEEK desirable when considering future radiographic studies. The primary benefits of this system include maintenance of lordotic angle and unaltered adjacent segment biomechanics. Disc and facet loading are also reduced. Due to its simple design, there is no need for additional screws or hardware. The surgical procedure is very simple and can be accomplished using minimally invasive techniques, which benefits future surgeries as well as reversal procedures.55 The only reported disadvantage is that flexion-extension and axial rotation (to some degree) are dampened by the implant.85 Other similar devices are the X-Stop™, DIAM™ and Coflex™ which are described below in detail.
The Coflex™ rivet system, shown in Figure 3-7, is an improvement over the earlier Coflex™ design that was a titanium device with a U-shaped body and wings at either end.55 The Coflex™ interspinous implant was invented by Dr. Jacques Samani in 1994 and has been in continuous use since 1995 outside the United States. This device restricted mobility in extension and rotation, while only permitting flexion. Additionally, like the Wallis™ system, these spacers did not compensate for instability during axial rotation and bending. Thus, the need for a device which will compensate for instability about all planes of motion drove the development of the Coflex™ rivet. This device was fixed to the bone using screws from right to left that pass through holes in the wings of the device and the spinous process. Results derived from a biomechanical study show that this device was able to compensate for instability in all modes of loading. It is noteworthy to point out that excessive unloading of the intervertebral disc is disadvantageous and physiologic loading-unloading should be maintained to sustain disc viability. This device is believed to allow for sufficient, but not excessive, amounts of load to be passed through the intervertebral disc.
The X-Stop™ system, shown in Figure 3-10, is an oval titanium alloy spacer with two lateral wings.55,86 While this device reduces extension, it allows motion during flexion, lateral bending and axial rotation. This device also enables the surgeon to perform a minimally invasive procedure and the entire surgery can be performed without general anesthesia. Studies have demonstrated that radiographic criteria such as foraminal height, width and cross-sectional area show greater improvement than with other interspinous spacers. The DIAM™ implant (Device for Intervertebral Assisted Motion) is made up of a silicon core encapsulated within a polyester sleeve.55,86 Its position is maintained by three bands, two across the spinous process and one around the supraspinous ligament.87
This device acts as a shock absorber and is not only designed for supporting the vertebra but also for distraction of the foramina. It functions as a pivot point for attaining balance and stability. It is a soft contact, compressible device that allows for uniform stress distribution across the bony joint.55,86 The flexion is controlled by decompression of the implant followed by the stretching of muscles, fascia and ligaments.
Biomechanical Evaluation of Dynamic Stabilization Systems
In vitro
A biomechanical experiment was conducted by Strauss et al.88 to compute the motion characteristics of a Graf Ligament™ implanted spine. They tested 13 cadaveric specimens, first in the intact mode, then injured and finally with the Graf Ligament™ system. Specimens were tested under pure moments of up to 10 Nm with a compressive preload of 500 N. Although ROM of the instrumented system was reduced in flexion-extension and lateral bending, axial rotation following instrumentation was 107% of intact ROM.
The Dynesys™ stabilization system has been widely studied. The first reported in vitro study was by Freudiger et al.89 in 1999. The authors reported a significant decrease in ROM in flexion-extension and horizontal translation and an increased vertical translation. This was attributed to the posterior shift of the COR. Furthermore, Schmoelz et al.90,91 compared Dynesys™ to a rigid fixation system and observed that Dynesys™ was comparable to the rigid instrumentation system in flexion, but allowed more motion in extension, lateral bending and axial rotation. No significant differences were reported in ROM and NZ at the adjacent levels. Intradiscal pressure (IDP) was measured in a subsequent work by the same investigators. Increased pressure was found in the case of flexion, while extension and lateral bending demonstrated a decrease in intradiscal pressure.91 Negative pressure readings were reported in extension, which demonstrated complete unloading of the disc. At the adjacent level, disc pressure was similar for both instrumented cases.90 The pressure increase in case of flexion was attributed to the posterior shift of the COR following implantation.92
Likewise, Niosi et al.93 investigated the effects of polymer spacer length on the quality of motion following Dynesys™ implantation. The Dynesys™ system restored the NZ of the injured specimen to a magnitude less than that of the intact spine. It was found that long spacers allowed for a larger ROM compared to spacers of shorter length. A key observation in this study was that tension of the (PET) cord of the device and spacer length played crucial roles in facet loading. Cheng et al.92 also studied the Dynesys™ device and found that it allowed significant axial rotation when implanted at two consecutive levels. Moreover, when combined with anterior fusion devices, Dynesys™ allowed greater ROM at the adjacent segments compared to rigid fixation.
An ideal dynamic system should function in a way that permits controlled ROM and increased load sharing with no effect on the stability of the construct. An Osteotech™ dynamic device was tested by Xu et al.94 and was compared with rigid instrumentation. Biomechanical testing involved 10 thoracolumbar specimens tested by applying pure moments of 10 Nm about all axes of loading. Specimens were tested intact, injured, following rigid stabilization and following dynamic device implantation. The dynamic device demonstrated greater ROM compared to the rigid system in flexion-extension and axial rotation.
Phillips et al.85 investigated the biomechanics of the DIAM™ device after facetectomy and discectomy. Six lumbar spines were used under the following conditions: intact, unilateral hemifacetectomy, discectomy and implanted with DIAM™. It was found that this device stabilized the motion segment and reduced segmental flexion/extension and lateral bending after combined facetectomy and discectomy to levels below that of the intact spine. Thus, DIAM™ was not capable of reducing axial rotation in the injured cases. The authors also suggested that the DIAM™ device can be utilized to dampen the altered kinematics of the adjacent levels following fusion.85 Wilke et al.95 performed a study in which four different interspinous implants, namely the Colfex™, Wallis™, DIAM™ and X-Stop™ were compared in terms of their flexibility and the intradiscal pressure. In extension mode, all the implants restricted the normal ROM by 50%. For all other loading modes, the ROM remained the same as the injured test case. The IDP was reduced significantly during extension but remained the same for all other loading modes. Despite their different designs all the devices were successful in unloading the disc during extension.
In silico
As mentioned previously, an FE model serves as an efficient tool for biomechanical analyses. For instance, a semi-rigid Twinflex™ system was analyzed by Templier et al.96 using an FE model of an L3-S1 segment. The goal of this work was to compare the load sharing characteristics of the Twinflex™ system versus a rigid posterior fixations system. It was found that a more homogeneous load transmission is achieved by reducing the flexural rigidity of the device. The authors observed that a rigid system might oppose the bending moment and lead to stress shielding, while a semi-rigid system will balance the moment by redistributing the loads though compressive and distractive forces. Additionally, Rohlmann et al.97 investigated a proposed device that was analogous to the Dynesys™ device. The group computed the ROM, intradiscal pressure, facet joint forces and implant stresses. The dynamic implant led to decreased intradiscal pressure, decreased facet forces and reduced ROM. In contrast, the rigid devices had a more pronounced effect. Still the differences between the dynamic and rigid systems were minimal. Both cases had almost no effect on the adjacent levels. Interestingly, Zander et al.98 investigated a similar device by placing the investigational device at the inferior adjacent level to a fusion. The implant was found to reduce the motion at the instrumented level except for axial rotation. Facet joint loads were also reduced, but intradiscal pressure was not affected by the device. This study, however, does not support the assumption that dynamic systems reduce the disc loads. The finite element method enables us to compute far more values than an experimental study and so is well suited for parametric studies. However, validation of these numerical models is mandatory, and, for this, experimental results are needed.
Due to instrumentation at one level, adjacent level disc tissue can be exposed to increased stresses. For instance, Castellvi et al.99 compared rigid versus dynamic Isobar TTL™ devices using a 3D FE model of the lumbar spine. The L5-S1 segment was fused and L4-L5 was implanted with a rigid or dynamic Isobar device. The dynamic system led to a reduction in maximum stresses at the adjacent level by a factor of 5.5% in flexion. On the other hand, the maximum stresses were increased by 17% at the instrumented level. It was also argued that the reduced rod stiffness may lead to increased axial rotation, thus resulting in reduced peak stresses with the COR shifted towards its physiologic location. Another parametric study on the stiffness of a generic dynamic stabilization device was performed by Shin et al.77 Using an FE model, three conditions were simulated: intact, fused segment (after facetectomy and laminectomy), and dynamic device stabilized (after facetectomy and laminectomy). ROM, adjacent and operative level disc pressure was computed. The dynamic device demonstrated more ROM compared to the fused condition and similar to that of the intact condition. Adjacent segment disc pressure was similar for intact and dynamically stabilized conditions, while the fused condition caused increased adjacent segment disc pressure. The optimum device stiffness was found to be 10-15 N/mm. Schmidt et al.100 carried out a similar study to understand the stiffness parameter of such devices and predicted that the direction of loading leads to a pairing of stiffness relations between axial rotation and lateral bending. Bending stiffness had a negligible effect in flexion and extension. Lateral bending and axial rotation were affected by both stiffness parameters. The stiffness values of these implants were demonstrated to be an important design parameter in terms of the designing/manufacturing of these devices for certain clinical indications.100
The major difference between the dynamic stabilization and arthroplasty is that dynamic stabilization aims at load sharing with the disc.35 Nevertheless, the popularity of dynamic stabilization systems is due to the lack of satisfaction with conventional fusion procedures rather than their proven superiority. Based on biomechanical studies and limited randomized trials it might be too early to comment on these devices.
NEWER TECHNOLOGIES AND FUTURE RESEARCH
Facet Replacement Systems
These are non-fusion spinal implants designed to treat spinal stenosis. Such devices replace the diseased facets in order to restore native motion and sagittal balance to the spine. The Archus TFAS™ system is one such ball-in-trough design with metal-on-metal articulations. The device is fixed by means of an interpedicular fixation system. Biomechanical testing demonstrated that following TFAS™ reconstruction, ROM is similar to that of the intact spine in all planes of loading, but the NZ was significantly increased in axial rotation.101 An FDA clinical trial is currently underway for this device. This trial seeks to gauge the restoration of stability and sagittal balance to the spine. Another notable device in this category is the AFRS™ (Anatomic Facet Replacement System). This precisely fit pedicle screw-based system is made using the CT scans of the facet joints in which the alignment of the implants is controlled by instrumentation. The implant allows up to ± 15 degrees of variability in the screw placement. The left and right anterior components are joined by cross-linking members for stability. The entire device is made of a cobalt-chrome-molybdenum alloy with hydroxyapatite-coated titanium screws. Biomechanical studies have demonstrated balanced facet loading with this device.55 FE as well as cadaveric data have confirmed that this device is able to restore stability. The biggest challenge among these devices is implant loosening, especially pedicle screw loosening under physiologic cyclic loading conditions.
Another notable device is the Total Posterior Facet replacement system (TOPS)™ system. It includes four polyaxial pedicle screws with an interlocking polycarbonate urethane (PCU) construct. The PCU element allows relative motion between the titanium plates and serves to limit motion.55 The shock absorptive nature of this implant allows controlled ROM by enabling constrained bending, straightening and twisting motions in order to restore the affected segment to the physiological NZ. The primary advantage that this system has over rigid systems is that the soft cushioning effect of the PCU minimizes screw-bone interface stresses. Due to limited clinical data that presently exists for this device, it is not reasonable to comment on the clinical efficacy of this device in terms of its ability to restore a physiologic ROM or even functional outcomes in the treatment of stenosis and spondylolisthesis.
360° Motion Preservation and Fusion Systems
This system is a combination of both anterior and posterior instrumentation. Specifically, an anterior artificial disc is matched up with a posterior dynamic stabilization device or facet replacement system. While disc replacement is often referred to as “total disc arthroplasty,” it is important to note that only the disc is being addressed by a “total” solution. The “three joint complex” (disc and two facet joints) is not addressed by total disc replacement.102
Recently, Goel et al.35 carried out an FE analysis of a 360° motion preservation system that constitutes both the artificial disc as well as the dynamic stabilization device. Their results suggest that this approach is capable of restoring normal ROM and may also address post-surgical complications resulting from TDA such as facet pain.
Shape Memory Alloys
Nitinol™ is an alloy with unique properties compared to most other metallic alloys. Its unique features are thermal shape memory and super elasticity. It has a distinctive property by which it has memory of its native shape and can return to that shape even after being deformed. This alloy can be used in spinal implants when the device must be implanted in a particular geometry, but a different geometry is required post-implantation.103
An implant that uses this technology is the Bioflex™ Spring Rod System (Fig. 3-9) in which a Nitinol™ rod is used in combination with pedicle screws across a spinal segment.55 Even though the system is designed to operate as a load-sharing element, it must also constrain the motion in flexion-extension. This device can also be used as a fusion device in combination with an interbody cage. Pre-tensioning of rods during surgery ensures that the semi-rigid device will perform in accordance with the physiologic loading conditions.55
Annuloplasty
Annular repair techniques are still considered a nascent technology. Current technology is pointing towards biological repair, as opposed to the biomechanical approaches that were discussed earlier in the chapter. The term annuloplasty includes prophylactic treatment post-microdiscectomy, treatment of fissures and tears in the disc as well as replication of connective tissue barriers and mechanical structures following implantation of a device.55 This technique might be used for the treatment of fissures and tears in the intervertebral disc, specifically attacking the early signs DDD. Growth factors and tissue engineering principles are currently being used to repair the degenerated disc tissues.
Tissue engineering of the intervertebral disc (IVD) will remain a key area of research for next several decades. Significant progress has already been made in the tissue engineering of an IVD. One of the key steps forward taken in the last few years has been by Dr. Robert Mauck’s group at Duke University. They used electrospinning technology to design a nanofibrous scaffold that allowed them to engineer the hierarchical structure of the annulus. Mesenchymal stem cells were seeded and a composite laminated lamellar structure was achieved after 10 weeks of in vitro culture.104-106 In order to be able to engineer both the central nucleus pulposus and the annulus fibrosus, a combination of agarose and nanofibrous scaffolds was used.106 Although anatomic features on a gross level were similar to native tissue, mechanical properties of the engineered disc were a fraction of that of the native disc.
As promising as such advances are, before tissue engineering delivers results in a clinical setting, several challenges must be overcome in three of its key components: cells, growth factors and scaffolds.104-106 Firstly, most cells used for IVD tissue engineering are from animals because human notochordal cells – cells that can develop into nucleus pulposus cells – exist only during embryonic development and during infancy. Consequently, data from such studies cannot be assumed to be feasible for humans.107 Key questions about the source of cells for disc repair are yet to be answered and consensus in the scientific community is missing. For instance, Luk and Ruan suggest that cell-based disc repair may not be suitable under severe disc degeneration.108 Secondly, growth factors like the transforming growth factor-b (TGF-b), fibroblast growth factor (FGF-2, bFGF and GDF-5), and bone morphogenetic proteins (BMP-2 and BMP-12) have been shown to have beneficial effects. However, further research is needed before the signaling cascade is fully understood. Finally, a wide variety of scaffolds are being experimented with to evaluate an optimal construct that allows engineering of discs with appropriate structure, biochemical signals and mechanical properties.107
Although major advances are being made in disc repair and regeneration, none of the current solutions restores physiological motion, spinal curvature and balance, IVD height and pressure, and appropriate load sharing.109 Also, not much attention has been paid to annulus closure.110
CONCLUSION
The future direction in spine surgery is towards minimally invasive techniques that promise shorter recovery times, reduced postoperative pain and shorter hospital stays. In contrast to the previous paradigms of rigid fixation, new technologies aim to restore and preserve motion while enabling a proper load sharing. In theory, proper load sharing and restoration of physiologic motion will reduce the probability of adjacent segment disease. Many of the devices described in this chapter are undergoing clinical trials; it will be premature to make strong predictions about the clinical efficacy of any of these devices on the basis of pre-clinical data owing to the fact that the spine is a complex system.
Current focusing of research efforts emphasizes long-term evaluation of devices and validation of theoretical and experimental benefits in a clinical setting. In addition to bench-top testing, well-designed, randomized clinical trials are needed to achieve these goals. Finally, proper patient selection and surgeon training are required for a successful clinical implementation of these novel technologies.
REFERENCES
- Kelsey JL, White AA 3rd. Epidemiology and impact of low-back pain. Spine (Phila Pa 1976). 1980;5(2):133-142.
- Toth JM, Wang M, Estes BT, Scifert JL, Seim HB 3rd, Turner AS. Polyetheretherketone as a biomaterial for spinal applications. Biomaterials. 2006;27(3):324-334.
- Schnake KJ, Putzier M, Haas NP, Kandziora F. Mechanical concepts for disc regeneration. Eur Spine J. 2006;15 Suppl 3:S354-360.
- Humphreys SC, Eck JC. Clinical evaluation and treatment options for herniated lumbar disc. Am Fam Physician. 1999;59(3):575-582, 587-588.
- Bush K, Cowan N, Katz DE, Gishen P. The natural history of sciatica associated with disc pathology. A prospective study with clinical and independent radiologic follow-up. Spine (Phila Pa 1976). 1992;17(10):1205-1212.
- Errico TJ. Lumbar disc arthroplasty. Clin Orthop Relat Res. 2005;(435):106-117.
- Feng Y, Egan B, Wang J. Genetic factors in intervertebral disc degeneration. Genes Dis. 2016;3(3):178-185.
- Iatridis JC, ap Gwynn I. Mechanisms for mechanical damage in the intervertebral disc annulus fibrosus. J Biomech. 2004;37(8):1165-1175.
- Goel VK, Monroe BT, Gilbertson LG, Brinckmann P. Interlaminar shear stresses and laminae separation in a disc. Finite element analysis of the L3-L4 motion segment subjected to axial compressive loads. Spine (Phila Pa 1976). 1995;20(6):689-698.
- Luoma K, Riihimäki H, Raininko R, Luukkonen R, Lamminen A, Viikari-Juntura E. Lumbar disc degeneration in relation to occupation. Scand J Work Environ Health. 1998;24(5):358-366.
- Mimura M, Panjabi MM, Oxland TR, Crisco JJ, Yamamoto I, Vasavada A. Disc degeneration affects the multidirectional flexibility of the lumbar spine. Spine (Phila Pa 1976). 1994;19(12):1371-1380.
- Vernon-Roberts B, Moore RJ, Fraser RD. The natural history of age-related disc degeneration: the pathology and sequelae of tears. Spine (Phila Pa 1976). 2007;32(25):2797-2804.
- Ala-Kokko L. Genetic risk factors for lumbar disc disease. Ann Med. 2002;34(1):42-47.
- Le Huec JC, Basso Y, Aunoble S, Friesem T, Bruno MB. Influence of facet and posterior muscle degeneration on clinical results of lumbar total disc replacement: two-year follow-up. J Spinal Disord Tech. 2005;18(3):219-223.
- Mooney V, Robertson J. The facet syndrome. Clin Orthop Relat Res. 1976;(115):149-156.
- Fujiwara A, Lim TH, An HS, et al. The effect of disc degeneration and facet joint osteoarthritis on the segmental flexibility of the lumbar spine. Spine (Phila Pa 1976). 2000;25(23):3036-3044.
- Fujiwara A, Tamai K, An HS, et al. The relationship between disc degeneration, facet joint osteoarthritis, and stability of the degenerative lumbar spine. J Spinal Disord. 2000;13(5):444-450.
- Goel VK, Gilbertson LG. Basic science of spinal instrumentation. Clin Orthop Relat Res. 1997;(335):10-31.
- Puttlitz CM, Goel VK, McLain RF. Biomechanics of spinal instrumentation. In: Chapman M, ed. Chapman’s Operative Orthopaedics. 2nd ed. Philadelphia, PA: Lippincott Raven Publishers; 2001: 3619-3632.
- Fraser RD. Interbody, posterior, and combined lumbar fusions. Spine (Phila Pa 1976). 1995;20(24 Suppl):167S-177S.
- Wang JM, Kim DJ, Yun YH. Posterior pedicular screw instrumentation and anterior interbody fusion in adult lumbar spondylolysis or grade I spondylolisthesis with segmental instability. J Spinal Disord. 1996;9(2):83-88.
- Yücesoy K, Yüksel KZ, Baek S, Sonntag VK, Crawford NR. Biomechanics of unilateral compared with bilateral lumbar pedicle screw fixation for stabilization of unilateral vertebral disease. J Neurosurg Spine. 2008;8(1):44-51.
- Ullrich PF Jr. Interbody cages for spine fusion. Spine Health. 2004. http://www.spine-health.com/treatment/spinal-fusion/interbody-cages-spine-fusion. Updated January 20, 2004.
- Spinal Fusion. NY Times. 2010. http://www.nytimes.com/slideshow/2007/08/01/health/100121Spinalfusionseries_5.html.
- Web. June 16, 2010.
- Wilke HJ, Kettler A, Goetz C, Claes L. Subsidence resulting from simulated postoperative neck movements: an in vitro investigation with a new cervical fusion cage. Spine (Phila Pa 1976). 2000;25(21):2762-2770.
- Gercek E, Arlet V, Delisle J, Marchesi D. Subsidence of stand-alone cervical cages in anterior interbody fusion: warning. Eur Spine J. 2003;12(5):513-516.
- Wilke HJ. Animal models for spinal research. Eur Spine J. 2008;17(1):1.
- Seller K, Wahl D, Wild A, Krauspe R, Schneider E, Linke B. Pullout strength of anterior spinal instrumentation: a product comparison of seven screws in calf vertebral bodies. Eur Spine J. 2007;16(7):1047-1054.
- Spivak JM, Neuwirth MG, Labiak JJ, Kummer FJ, Ricci JL. Hydroxyapatite enhancement of posterior spinal instrumentation fixation. Spine (Phila Pa 1976). 1994;19(8):955-964.
- Yerby SA, Ehteshami JR, McLain RF. Offset laminar hooks decrease bending moments of pedicle screws during in situ contouring. Spine (Phila Pa 1976). 1997;22(4):376-381.
- Hsu CC, Chao CK, Wang JL, Hou SM, Tsai YT, Lin J. Increase of pullout strength of spinal pedicle screws with conical core: biomechanical tests and finite element analyses. J Orthop Res. 2005;23(4):788-794.
- Brantigan JW, Steffee AD, Geiger JM. A carbon fiber implant to aid interbody lumbar fusion. Mechanical testing. Spine (Phila Pa 1976). 1991;16(6 Suppl):S277-282.
- Schleicher P, Gerlach R, Schär B, et al. Biomechanical comparison of two different concepts for stand alone anterior lumbar interbody fusion. Eur Spine J. 2008;17(12):1757-1765.
- Wittenberg RH, Shea M, Edwards WT, Swartz DE, White AA 3rd, Hayes WC. A biomechanical study of the fatigue characteristics of thoracolumbar fixation implants in a calf spine model. Spine (Phila Pa 1976). 1992;17(6 Suppl):S121-128.
- Goel VK, Kiapour A, Faizan A, Krishna M, Friesem T. Finite element study of matched paired posterior disc Implant and dynamic stabilizer (360° motion preservation system). SAS J. 2007;1(1):55-61.
- Grosland NM, Goel VK. Vertebral endplate morphology follows bone remodeling principles. Spine (Phila Pa 1976). 2007;32(23):E667-673.
- Ivanov A, Kiapour A, Ebraheim N, Goel VK. Simulation of the transverse fracture of the sacrum using a finite element model of lumbar spine-pelvis segment. American Society of Mechanical Engineers Summer Bioengineering Conference (SBC); Marco Island, FL: June 2008.
- Rohlmann A, Bauer L, Zander T, Bergmann G, Wilke HJ. Determination of trunk muscle forces for flexion and extension by using a validated finite element model of the lumbar spine and measured in vivo data. J Biomech. 2006;39(6):981-989.
- Shirazi-Adl A, Drouin G. Load-bearing role of facets in a lumbar segment under sagittal plane loadings. J Biomech. 1987;20(6):601-613.
- Polikeit A, Ferguson SJ, Nolte LP, Orr TE. Factors influencing stresses in the lumbar spine after the insertion of intervertebral cages: finite element analysis. Eur Spine J. 2003;12(4):413-420.
- Sasa T, Yoshizumi Y, Imada K, et al. Cervical spondylolysis in a judo player: a case report and biomechanical analysis. Arch Orthop Trauma Surg. 2009;129(4):559-567.
- Grosland NM, Goel VK, Grobler LJ. Comparative biomechanical investigation of multiple interbody fusion cages: a finite element analysis. Proceedings of the 45th Annual Meeting of the Orthopaedic Research Society; Anaheim, CA: 1999.
- Zhang QH, Tan SH, Chou SM. Investigation of fixation screw pull-out strength on human spine. J Biomech. 2004;37(4):479-485.
- McAfee PC, Farey ID, Sutterlin CE, Gurr KR, Warden KE, Cunningham BW. The effect of spinal implant rigidity on vertebral bone density. A canine model. Spine (Phila Pa 1976). 1991;16(6 Suppl):S190-197.
- Hojo Y, Kotani Y, Ito M, et al. A biomechanical and histological evaluation of a bioresorbable lumbar interbody fusion cage. Biomaterials. 2005;26(15):2643-2651.
- Gurr KR, McAfee PC, Shih CM. Biomechanical analysis of anterior and posterior instrumentation systems after corpectomy. A calf-spine model. J Bone Joint Surg Am. 1988;70(8):1182-1191.
- Hilibrand AS, Robbins M. Adjacent segment degeneration and adjacent segment disease: the consequences of spinal fusion? Spine J. 2004;4(6 Suppl):190S-194S.
- Okuda S, Iwasaki M, Miyauchi A, Aono H, Morita M, Yamamoto T. Risk factors for adjacent segment degeneration after PLIF. Spine (Phila Pa 1976). 2004;29(14):1535-1540.
- Park P, Garton HJ, Gala VC, Hoff JT, McGillicuddy JE. Adjacent segment disease after lumbar or lumbosacral fusion: review of the literature. Spine (Phila Pa 1976). 2004;29(17):1938-1944.
- Throckmorton TW, Hilibrand AS, Mencio GA, Hodge A, Spengler DM. The impact of adjacent level disc degeneration on health status outcomes following lumbar fusion. Spine (Phila Pa 1976). 2003;28(22):2546-2550.
- Sekhon LH, Ball JR. Artificial cervical disc replacement: principles, types and techniques. Neurol India. 2005;53(4):445-450.
- Gamradt SC, Wang JC. Lumbar disc arthroplasty. Spine J. 2005;5(1):95-103.
- Valdevit A, Errico TJ. Design and evaluation of the FlexiCore metal-on-metal intervertebral disc prosthesis. Spine J. 2004;4(6 Suppl):276S-288S.
- Bono CM, Garfin SR. History and evolution of disc replacement. Spine J. 2004;4(6 Suppl):145S-150S.
- Kim D, Sengupta DK, Cammisa FC Jr, Yoon DH, Fessley RG, eds. Dynamic Reconstruction of the Spine. 2nd ed. New York, NY; Thieme Medical Publishers, Inc.: 2015.
- Kaner T, Sasani M, Oktenoglu T, Ozer AF. Dynamic stabilization of the spine: a new classification system. Turk Neurosurg. 2010;20(2):205-215.
- Santos EG, Polly DW Jr, Mehbod AA, Saleh KJ. Disc arthroplasty: lessons learned from total joint arthroplasty. Spine J. 2004;4(6 Suppl):182S-189S.
- Demetropoulos CK, Sengupta DK, Knaub MA, et al. Biomechanical evaluation of the kinematics of the cadaver lumbar spine following disc replacement with the ProDisc-L prosthesis. Spine (Phila Pa 1976). 2010;35(1):26-31.
- Hitchon PW, Eichholz K, Barry C, et al. Biomechanical studies of an artificial disc implant in the human cadaveric spine. J Neurosurg Spine. 2005;2(3):339-343.
- Cunningham BW, Gordon JD, Dmitriev AE, Hu N, McAfee PC. Biomechanical evaluation of total disc replacement arthroplasty: an in vitro human cadaveric model. Spine (Phila Pa 1976). 2003;28(2):S110-117.
- Ha SK, Kim SH, Kim DH, Park JY, Lim DJ, Lee SK. Biomechanical study of lumbar spinal arthroplasty with a semi-constrained artificial disc (activ L) in the human cadaveric spine. J Korean Neurosurg Soc. 2009;45(3):169-175.
- Serhan HA, Dooris AP, Parsons ML, Ares PJ, Gabriel SM. In vitro wear assessment of the Charité Artificial Disc according to ASTM recommendations. Spine (Phila Pa 1976). 2006;31(17):1900-1910.
- Anderson PA, Rouleau JP, Toth JM, Riew KD. A comparison of simulator-tested and -retrieved cervical disc prostheses. Invited submission from the Joint Section Meeting on Disorders of the Spine and Peripheral Nerves, March 2004. J Neurosurg Spine. 2004;1(2):202-210.
- Anderson PA, Sasso RC, Rouleau JP, Carlson CS, Goffin J. The Bryan Cervical Disc: wear properties and early clinical results. Spine J. 2004;4(6 Suppl):303S-309S.
- Paré PE, Chan FW, Powell ML. Wear characterization of the A-MAV™ anterior motion replacement using a spine wear simulator. WEAR. 2007;263(7-12):1055-1059.
- Dooris AP, Goel VK, Grosland NM, Gilbertson LG, Wilder DG. Load-sharing between anterior and posterior elements in a lumbar motion segment implanted with an artificial disc. Spine (Phila Pa 1976). 2001;26(6):E122-129.
- Denozière G, Ku DN. Biomechanical comparison between fusion of two vertebrae and implantation of an artificial intervertebral disc. J Biomech. 2006;39(4):766-775.
- Goel VK, Grauer JN, Patel TCh, et al. Effects of charité artificial disc on the implanted and adjacent spinal segments mechanics using a hybrid testing protocol. Spine (Phila Pa 1976). 2005;30(24):2755-2764.
- Le Huec JC, Lafage V, Bonnet X, et al. Validated finite element analysis of the maverick total disc prosthesis. J Spinal Disord Tech. 2010;23(4):249-257.
- Rawlinson JJ, Punga KP, Gunsallus KL, Bartel DL, Wright TM. Wear simulation of the ProDisc-L disc replacement using adaptive finite element analysis. J Neurosurg Spine. 2007;7(2):165-173.
- Goreham-Voss CM, Hyde PJ, Hall RM, Fisher J, Brown TD. Cross-shear implementation in sliding-distance-coupled finite element analysis of wear in metal-on-polyethylene total joint arthroplasty: intervertebral total disc replacement as an illustrative application. J Biomech. 2010;43(9):1674-1681.
- Bhattacharya S, Goel VK, Liu X, Kiapour A, Serhan H. Models that incorporate spinal structures predict better wear performance of cervical artificial discs. Spine J. 2011;11(8):766-776.
- Taylor BA, Okubadejo GO, Patel AA, et al. Evaluation of total disc arthroplasty: a canine model. Am J Orthop (Belle Mead NJ). 2008;37(4):E64-70.
- McAfee PC, Cunningham BW, Orbegoso CM, Sefter JC, Dmitriev AE, Fedder IL. Analysis of porous ingrowth in intervertebral disc prostheses: a nonhuman primate model. Spine (Phila Pa 1976). 2003;28(4):332-340.
- Cunningham BW, Dmitriev AE, Hu N, McAfee PC. General principles of total disc replacement arthroplasty: seventeen cases in a nonhuman primate model. Spine (Phila Pa 1976). 2003;28(20):S118-124.
- Cunningham BW. Basic scientific considerations in total disc arthroplasty. Spine J. 2004;4(6 Suppl):219S-230S.
- Shin DS, Lee K, Kim D. Biomechanical study of lumbar spine with dynamic stabilization device using finite element method. Computer-aided design. 2007;39(7):559-567.
- Galbusera F, Bellini CM, Zweig T, et al. Design concepts in lumbar total disc arthroplasty. Eur Spine J. 2008;17(12):1635-1650.
- Tortolani PJ, Cunningham BW, Eng M, McAfee PC, Holsapple GA, Adams KA. Prevalence of heterotopic ossification following total disc replacement. A prospective, randomized study of two hundred and seventy-six patients. J Bone Joint Surg Am. 2007;89(1):82-88.
- Mulholland RC, Sengupta DK. Rationale, principles and experimental evaluation of the concept of soft stabilization. Eur Spine J. 2002;11 Suppl 2:S198-S205.
- Nockels RP. Dynamic stabilization in the surgical management of painful lumbar spinal disorders. Spine (Phila Pa 1976). 2005;30(16 Suppl):S68-72.
- Schwarzenbach O, Berlemann U, Stoll TM, Dubois G. Posterior dynamic stabilization systems: DYNESYS. Orthop Clin North Am. 2005;36(3):363-372.
- Sénégas J, Vital JM, Pointillart V, Mangione P. Clinical evaluation of a lumbar interspinous dynamic stabilization device (the Wallis system) with a 13-year mean follow-up. Neurosurg Rev. 2009;32(3):335-341.
- Sengupta DK. Dynamic stabilization devices in the treatment of low back pain. Neurol India. 2005;53(4):466-474.
- Phillips FM, Voronov LI, Gaitanis IN, Carandang G, Havey RM, Patwardhan AG. Biomechanics of posterior dynamic stabilizing device (DIAM) after facetectomy and discectomy. Spine J. 2006;6(6):714-722.
- Sobottke R, Schlüter-Brust K, Kaulhausen T, et al. Interspinous implants (X Stop, Wallis, Diam) for the treatment of LSS: is there a correlation between radiological parameters and clinical outcome? Eur Spine J. 2009;18(10):1494-1503.
- Phillips FM, Voronov LI, Gaitanis IN, Carandang G, Havey RM, Patwardhan AG. Biomechanics of posterior dynamic stabilizing device (DIAM) after facetectomy and discectomy. Spine J. 2006;6(6):714-722.
- Strauss PJ, Novotny JE, Wilder DG, Grobler LJ, Pope MH. Multidirectional stability of the Graf system. Spine (Phila Pa 1976). 1994;19(8):965-972.
- Freudiger S, Dubois G, Lorrain M. Dynamic neutralisation of the lumbar spine confirmed on a new lumbar spine simulator in vitro. Arch Orthop Trauma Surg. 1999;119(3-4):127-132.
- Schmoelz W, Huber JF, Nydegger T, Dipl-Ing, Claes L, Wilke HJ. Dynamic stabilization of the lumbar spine and its effects on adjacent segments: an in vitro experiment. J Spinal Disord Tech. 2003;16(4):418-423.
- Schmoelz W, Huber JF, Nydegger T, Claes L, Wilke HJ. Influence of a dynamic stabilisation system on load bearing of a bridged disc: an in vitro study of intradiscal pressure. Eur Spine J. 2006;15(8):1276-1285.
- Cheng BC, Gordon J, Cheng J, Welch WC. Immediate biomechanical effects of lumbar posterior dynamic stabilization above a circumferential fusion. Spine (Phila Pa 1976). 2007;32(23):2551-2557.
- Niosi CA, Zhu QA, Wilson DC, Keynan O, Wilson DR, Oxland TR. Biomechanical characterization of the three-dimensional kinematic behaviour of the Dynesys dynamic stabilization system: an in vitro study. Eur Spine J. 2006;15(6):913-922.
- Xu HZ, Wang XY, Chi YL, et al. Biomechanical evaluation of a dynamic pedicle screw fixation device. Clin Biomech (Bristol, Avon). 2006;21(4):330-336.
- Wilke HJ, Drumm J, Häussler K, Mack C, Steudel WI, Kettler A. Biomechanical effect of different lumbar interspinous implants on flexibility and intradiscal pressure. Eur Spine J. 2008;17(8):1049-1056.
- Templier A, Denninger L, Mazel C, Lavaste F, Skalli W. Comparison between two different concepts of lumbar posterior osteosynthesis implants. Eur J Orthop Surg Traumatology. 1998;8(1):27-36.
- Rohlmann A, Burra NK, Zander T, Bergmann G. Comparison of the effects of bilateral posterior dynamic and rigid fixation devices on the loads in the lumbar spine: a finite element analysis. Eur Spine J. 2007;16(8):1223-1231.
- Zander T, Rohlmann A, Burra NK, Bergmann G. Effect of a posterior dynamic implant adjacent to a rigid spinal fixator. Clin Biomech (Bristol, Avon). 2006;21(8):767-774.
- Castellvi A. Stress reduction in adjacent level discs via dynamic instrumentation: a finite element analysis. SAS J. 2007;1(2):74-81.
- Schmidt H, Heuer F, Wilke HJ. Which axial and bending stiffness of posterior implants are required to design a flexible lumbar stabilization system? J Biomech. 2009;42(1):48-54.
- Serhan HA, Varnavas G, Dooris AP, Patwadhan A, Tzermiadianos M. Biomechanics of the posterior lumbar articulating elements. Neurosurg Focus. 2007;22(1):E1.
- Kiapour A, Mhatre D, Koruprolu S, et al. A 360 posterior dynamic stabilization system restores the kinematics of a lumbar segment to normal: in vitro & FEM study. In: 55th Annual Meeting of the Orthopaedic Research Society. Las Vegas, NV, 2009.
- Ruttmann E, Laufer G, Müller, L. Development of a minimally invasive mitral valve surgery program – The Innsbruck experience. Eur Surg. 2006;38:320-325.
- Nerurkar NL, Baker BM, Sen S, Wible EE, Elliott DM, Mauck RL. Nanofibrous biologic laminates replicate the form and function of the annulus fibrosus. Nat Mater. 2009;8(12):986-992.
- Nerurkar NL, Elliott DM, Mauck RL. Mechanics of oriented electrospun nanofibrous scaffolds for annulus fibrosus tissue engineering. J Orthop Res. 2007;25(8):1018-1028.
- Nerurkar NL, Sen S, Huang AH, Elliott DM, Mauck RL. Engineered disc-like angle-ply structures for intervertebral disc replacement. Spine (Phila Pa 1976). 2010;35(8):867-873.
- Yang X, Li X. Nucleus pulposus tissue engineering: a brief review. Eur Spine J. 2009;18(11):1564-1572.
- Luk KD, Ruan DK. Intervertebral disc transplantation: a biological approach to motion preservation. Eur Spine J. 2008;17 Suppl 4:504-510.
- Leung VY, Chan D, Cheung KM. Regeneration of intervertebral disc by mesenchymal stem cells: potentials, limitations, and future direction. Eur Spine J. 2006;15 Suppl 3:S406-413.
- Bron JL, van der Veen AJ, Helder MN, et al. Biomechanical and in vivo evaluation of experimental closure devices of the annulus fibrosus designed for a goat nucleus replacement model. Eur Spine J. 2010;19(8):1347-1355.