Satoru Demura*, Katsuhito Yoshioka† and Satoshi Kato‡
TREATMENT STRATEGY FOR SPINAL METASTASES*
In the past, the indication for surgical management of spinal metastases was based on signs of neurological deficit, presence of intractable pain, and spinal instability. These indications were described by Harrington et al. with a five-category classification for spinal metastases based on the neurologic compromise and vertebral collapse and/or instability:1
| Class I: | No significant neurologic involvement. |
| Class II: | Involvement of bone without collapse or instability. |
| Class III: | Major neurologic impairment (sensory or motor) without significant involvement of bone. |
| Class IV: | Vertebral collapse with pain resulting from mechanical causes or instability, but with no significant neurologic compromise. |
| Class V: | Vertebral collapse or instability combined with major neurologic impairment. |
He recommended that patients with class I, II and III can be treated non-surgically with chemotherapy, hormonal manipulation and/or local irradiation. Class IV or V is an indication for surgical intervention.
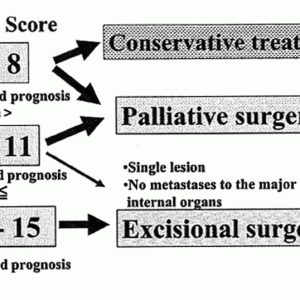
Tokuhashi et al. originally reported a scoring system for preoperative evaluation of a patient’s prognosis with metastatic spine tumors.2 They revised their scoring system and evaluated treatment outcomes using their strategy.3,4 Six factors were included: general medical condition, number of extraspinal bone metastases, number of vertebral metastases, visceral metastases, primary site of the cancer, and presence of neurological deficit. Each parameter is evaluated with a score, giving a maximum total score of 15. Excisional surgery is indicated with a total score of 12 or more, whereas a palliative surgery would be recommended for a score of 9 to 11. In patients with a total score of 8 or less, conservative or palliative procedures will be indicated (Table 2-1, Fig. 2-1).
| Characteristic | Score | |
|---|---|---|
| General condition (performance status) | ||
| Poor (PS 10%-40%) | 0 | |
| Moderate (PS 50%-70%) | 1 | |
| Good (PS 80%-100%) | 2 | |
| No. of extraspinal bone metastases foci | ||
| ≥3 | 0 | |
| 1-2 | 1 | |
| 0 | 2 | |
| No. of metastases in the vertebral body | ||
| ≥3 | 0 | |
| 2 | 1 | |
| 1 | 2 | |
| Metastases to the major internal organs | ||
| Unremovable | 0 | |
| Removable | 1 | |
| No metastases | 2 | |
| Primary site of the cancer | ||
| Lung, osteosarcoma, stomach, bladder, esophagus, pancreas | 0 | |
| Liver, gallbladder, unidentified | 1 | |
| Others | 2 | |
| Kidney, uterus | 3 | |
| Rectum | 4 | |
| Thyroid, breast, prostate, carcinoid tumor | 5 | |
| Palsy | ||
| Complete (Frankel A, B) | 0 | |
| Incomplete (Frankel C, D) | 1 | |
| None (Frankel E) | 2 | |
| Criteria of predicted prognosis: Total Score (TS) 0-8 = >6 mo; TS 9-11 = ≤6 mo; TS 12-15 = ≤1 yr. | ||
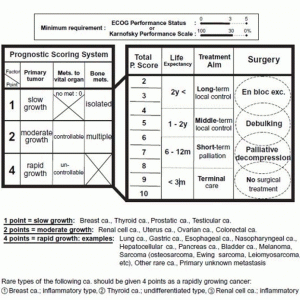
Tomita et al. proposed a surgical strategy for spinal metastases which might be used to select not only whether a patient is a candidate for surgical intervention, but to guide the selection of surgery. They showed the three most important prognostic factors: grade of malignancy of the primary tumor, visceral metastases to vital organs, and bone metastases.5,6 These three factors are added together to give a total prognostic score between 2 and 10 points. The treatment aim for each patient is set according to this prognostic score and life expectancy (Fig. 2-2). In the case of surgical treatment, ECOG (Eastern Cooperative Oncology Group) performance status (at least less than 3) or Karnofsky performance scale (more than 30%) are also required, which are the same requirements for administering chemotherapy.
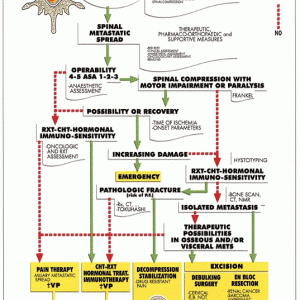
Cappuccio et al. showed a flow-chart for the treatment of spinal metastases.7 This chart accounts for various factors associated with spinal metastases, such as: spread of spinal metastases, anesthetic assessment, presence of spinal cord compression with neurological deficit, the risk of pathological fracture, the sensitivity to adjuvant therapy, number of vertebral metastases, and therapeutic possibilities in osseous and/or visceral metastases. When the treatment was selected by the proposed algorithm, the majority of the patients have a good residual quality of life in their retrospective review (Fig. 2-3).
Fisher et al. proposed a spine instability neoplastic score (SINS) based on patient symptoms and radiographic criteria of the spine.8,9 The classification system was developed by systematic reviews, expert opinion on the Delphi technique. The components consist of tumor location, mechanical pain, bone lesion quality, spinal alignment, vertebral body collapse, and posterolateral involvement of spinal elements. These six factors are combined to give a minimum score of 2 and maximum score of 18 (Table 2-2). They also confirmed highly inter- and intraobserver reliability.
| Element of SINS | Score | |
| Location | ||
| Junctional (occiput-C2, C7-T2, T11-L1, L5-S1) | 3 | |
| Mobile spine (C3-C6, L2-L4) | 2 | |
| Semi-rigid (T3-T10) | 1 | |
| Rigid (S2-S5) | 0 | |
| Pain relief with recumbency and/or pain with movement/loading of the spine | ||
| Yes | 3 | |
| No (occasional pain but not mechanical) | 1 | |
| Pain free lesion | 0 | |
| Bone lesion | ||
| Lytic | 2 | |
| Mixed (lytic/blastic) | 1 | |
| Blastic | 0 | |
| Radiographic spinal alignment | ||
| Subluxation/translation present | 4 | |
| De novo deformity (kyphosis/scoliosis) | 2 | |
| Normal alignment | 0 | |
| Vertebral body collapse | ||
| >50% collapse | 3 | |
| <50% collapse | 2 | |
| No collapse with >50% body involved | 1 | |
| None of the above | 0 | |
| Posterolateral involvement of the spinal elements (facet, pedicle or CV joint fracture or replacement with tumor) | ||
| Bilateral | 3 | |
| Unilateral | 1 | |
| None of the above | 0 | |
SURGICAL CLASSIFICATION OF SPINE TUMORS*
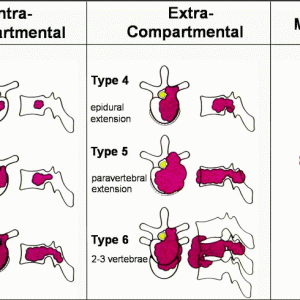
Based on the pattern of local vertebral tumor progression and the type of surgery used, Tomita et al. devised a “Surgical classification of spinal tumors.”5,6 Metastatic tumors frequently grow in the middle-posterior part of the vertebral body, from where they can extend through the pedicles (type 1, 2, 3). Then, the tumor generally grows outside the compartment and into the spinal canal (type 4), or extends outside of the vertebra (type 5), and finally to the adjacent vertebrae (type 6) (Fig. 2-4). Boriani et al. proposed the surgical staging for spine tumor. In the transverse plane, the vertebra is divided into 12 zones for the planning of surgical procedures and reliable information between institutions.10 (see Section 20 Chapter 1)
TREATMENT OPTIONS FOR SPINAL METASTASES*
Surgery
The selection of the surgical treatment for each patient is considered according to their life expectancy and the pattern of vertebral tumor progression. En bloc excision, such as total en bloc spondylectomy, is recommended for patients with technically resectable tumors and an expected good/satisfactory long-term prognosis. When the patients are expecting middle-term survival, it is desirable to excise the tumor as much as possible (debulking) to prevent neurological deficit by local recurrence. Palliative decompression and stabilization of the spinal column is desirable for patients with a predicted prognosis of 6 to 12 months. Recently, minimally invasive stabilization using percutaneous pedicle screws with/without decompression is being used as another treatment for spinal metastatic patients.11
Vertebroplasty and Kyphoplasty
Vertebroplasty, the injection of polymethylmethacrylate (PMMA) cement into a compromised vertebral segment, stabilizes and strengthens the anterior column. Unlike the delayed benefit of radiation, symptom relief in vertebroplasty is often immediate. Kyphoplasty is a technique designed to reduce deformity and spinal malalignment. The potential risks of the procedure include cement leakage, spinal cord compression associated with dorsal cement extravasation, embolization of the vessels, and vertebral body collapse.12-14
Radiotherapy
Radiotherapy is a well-recognized, effective palliative treatment for spinal metastases. General indications for radiotherapy are pain without instability, neurologic compromise, and/or deformity. For symptomatic patients with extensive metastases, irradiation to the spine may be appropriate. The most common and conventional dose is 30 Gy in 10 fractions.15 Because preoperative radiation makes it difficult to dissect around dura due to adhesion during surgery and increases the rate of surgical site infection and cerebrospinal fluid leakage, preoperative radiation should be avoided if possible.16,17 Intraoperative radiation treatment of spine tumors has been explored using a single-dose, custom-designed electron beam that minimizes collateral damage through precision targeting and can be applied in tandem with traditional external-beam radiotherapy.18
Instead of conventional radiotherapy, stereotactic radiotherapy has evolved in an attempt to enhance single-fraction doses to a localized tumor while avoiding radiation-induced myelopathy. Excellent clinical improvement of pain and neurologic function has been reported, especially in patients with solitary spinal metastases, to increase the prospects of long-term local control.19,20
Bisphosphonate and Denosumab
The use of bisphosphonate for spinal metastases has been considered to prevent or delay skeletal-related complications including pathologic compression fractures and spinal cord compression. Several types of bisphosphonates with different activation mechanisms have been developed. Zoledronic acid inhibits the bone resorption through osteoclasts, which then induce apoptosis. It has been reported to be effective for treating a variety of cancers with bone metastases.21,22 Denosumab also has been available in the treatment of bone metastases. It suppresses osteoclast activation mediated by RANKL inhibition. Current reports have shown that denosumab had a greater effect compared with zoledronic acid, especially in patients with breast or prostate cancer.23
BREAST CANCER†
When compared to other metastatic spinal tumors, patients with breast cancer have a relatively long life expectancy. Over the years, the prognosis of breast cancer has improved following advances in pharmacologic and surgical techniques in controlling regional disease. However, metastatic progression involving distal sites such as bone, lung, liver, and brain remains common.24,25 Despite multimodal treatment, approximately 20% to 30% of women with early-stage breast cancer still experience distant metastases.26,27 Bone is the most frequent site of metastases in breast cancer patients. Breast cancer metastases constitute the most common cause of symptomatic spine metastases, accounting for 9%–40% of reported clinical series on spinal epidural metastases in the literature.28,29 Pure osteolytic lesions are most common, followed by mixed osteolytic lesions. However, in 10% to 15% of metastases, the lesions are purely blastic.30 Patients with breast cancer are also at higher risk for vertebral compression fractures because of age, osteoporosis, and the osteolytic nature of the tumor. Chemotherapy-induced early menopause causes rapid bone loss and may increase the risk of osteoporosis later in life.31,32 Breast tumors with bone metastases have been shown to be more frequently estrogen receptor-positive and better differentiated than breast tumors with visceral metastases. Estrogen receptor-negative tumors are associated with a comparatively higher proliferative rate, visceral metastases, and a generally poorer prognosis. Large tumor size, presence of visceral metastases, and associated neoplastic syndrome (e.g., hypercalcemia) worsen prognosis.33 Inflammatory breast carcinoma is characterized by an early age at diagnosis, poor nuclear grade, negative hormone receptor status, and generally poor survival outcome. It appears to be a relatively rare form with distinct clinicopathologic features.34,35
The optimum treatment algorithm has not been definitively defined, with available options including pharmacologic management, radiotherapy, and surgery. The exact surgical indications for patients with metastatic breast cancer to the spine are controversial, and evidence-based guidelines are not available due to paucity of the literature on this topic. Early management of metastatic spinal tumors traditionally emphasized treatment with radiotherapy over surgical decompression. Most commonly, patients are treated with radiotherapy.36,37 Radiotherapy is capable of promoting re-ossification, leading to increased stability of spinal metastases. Strategies to reduce risk of bone metastases are now undergoing clinical testing. Oral clodronate, a bisphosphonate, reduced risk of new bone metastases by 50% in a randomized trial on high-risk breast cancer patients followed for a median of 36 months.38 Bisphosphonates are effective in normalizing hypercalcemia, reducing the pain associated with bone metastases, and decreasing the risk of pathologic fractures. Combined therapy with a bisphosphonate and a hormone antagonist is commonly recommended for patients with bone metastases. Surgical treatment may be indicated in cases with one or more of the following criteria: 1) progressive neurologic deficit; 2) instability of the spine due to pathologic fracture causing intractable pain; 3) single spinal lesion; 4) prior irradiation of the site of progressive spinal involvement with cord compression; 5) life expectancy longer than 3 months; and 6) responsive or stable disease in other metastatic sites.29,39 If radical excision with wide or marginal margin such as total en bloc spondylectomy is performed for an isolated spinal metastasis, local control may be achievable in the long term. The treatment of breast cancer keeps changing, and surgical treatment is considered one essential option among the existing treatment modalities.
PROSTATE CANCER†
Prostate cancer metastases to the spine are common. The most common radiographic appearance is osteoblastic, while some cases present a mixed lytic and blastic appearance, and osteolytic lesions are rare. Since these metastatic lesions are usually blastic, pathologic fractures are relatively rare. Also, neurologic involvement is uncommon for blastic lesions. Treatment of prostate cancer involves different modalities such as chemotherapy, radiation, hormonal manipulation, and surgery. Although prostate cancer patients with metastatic spinal cord compression have a relatively longer life expectancy compared to other tumors, the neurologic complication has a relevant impact on survival.40-43
Hormone withdrawal, whether by bilateral orchidectomies or androgen deprivation, is initially effective in relieving symptoms in 70 to 80% of patients. However, the tumors frequently acquire resistance to hormone manipulation. Response rates to chemotherapy are less than 20% in most series, and chemotherapy has been shown to have little effect on overall survival in advanced disease. In contrast, radiation therapy provides effective palliation for spine metastases. Recently, new, highly effective systemic agents have significantly changed the treatment landscape for castration-resistant prostate cancer patients, improving life expectancy.44 Some of these therapies have shown documented efficacy in delaying skeletal-related events and improving bone pain.
Abnormal osteoclast activity is the rationale for administration of potent osteolysis inhibitors such as zoledronic acid and denosumab. Bisphosphonates have direct influence on osteoclasts and also appear to inhibit the invasion, adhesion, and migration of prostate cancer cells.45 Denosumab produced a 33% reduction to risk of symptomatic bone metastasis.46 These agents reduce the burden of bone metastatic disease, although such benefit does not translate to an improvement in survival.47
Surgical treatment of prostate cancer metastasis to the spine is generally limited. Surgery has been proposed as reasonable palliative alternative for patients.48 Therefore, only a few studies specifically address surgical treatment of metastatic spinal cord compression in prostate cancer.49-52 Surgical treatment was performed in cases with one or more of the following: 1) obvious spinal deformity with intractable pain; 2) prior irradiation of the site of progressive spinal involvement; and 3) medically intractable mechanical pain or neurological deficit. These selection criteria for surgical intervention in prostate cancer spinal metastases, however, may be limited by a deteriorating general condition in patients deemed medically unstable to tolerate proposed surgery.
LUNG CANCER†
Lung cancer is considered the most common cancer worldwide. Lung cancer commonly spreads to the liver, the lymph nodes, adrenals, bones, and brain. Approximately 65% of patients with lung cancer develop bone metastases. For patients with non-small cell lung cancer (NSCLC), the spinal column is the most common site of bone metastasis.53
In general, majority of these patients are treated with systemic therapy as a symptoms-based palliative approach. Patients with spinal metastasis from lung cancer are often not candidates for surgical treatment, because 1) survival is limited at the time of diagnosis; and 2) spinal metastasis is usually multiple at the time of detection.54 For patients with a short life expectancy, less invasive interventions such as kyphoplasty, vertebroplasty, and/or radiotherapy are advocated irrespective of whether the patient has neurologic deficit/s.
However, Morgen et al. found a statistically significant increase in survival over the years for lung cancer patients with metastatic spinal cord compression (MSCC).55 In their patients with lung cancer who underwent surgery for MSCC, 1-year survival increased from 9% in year 2005 to 30% in year 2010.55 Improvements in adjuvant treatment modalities including chemotherapy, radiotherapy, small molecule inhibitors, and immunotherapies have resulted in increased control of systemic disease and longer life expectancies. The improvement in prognosis could expand the role of surgical treatment. Several studies have reported positive results for surgical intervention in metastatic spinal cord compression cases from NSCLC.56-60 Even for patients with advanced metastatic lung cancer, some may experience long-term benefit from surgical intervention. Recently, en bloc resection of spinal metastasis has been reported to show promising results in selected cases with controllable primary lung cancer, localized spinal metastasis, and no visceral metastasis.54 Synchronous resection of solitary bone metastasis and lung cancer resection has been reported as a potential therapeutic option.61 However, strict patient selection is required because of the potential risk of complications.
KIDNEY CANCER‡
The bone is a common site of metastatic renal cell carcinoma (RCC), second only to the lung, and most often involves the spine.62,63 Spinal metastases originating from RCC are difficult to manage. They tend to be large, highly destructive, hypervascular, and more resistant to systemic and radiation therapy than other metastases, resulting in pathologic fractures and spinal cord compression that severely compromise the performance status and quality of life of the patients. The severity of the skeletal related events (SREs) in RCC is well-illustrated by the recent study by Woodward et al.64 in which the mean number of SREs experienced by the bone metastatic patients over the course of their disease was 2.4 and only 37/254 patients experienced no skeletal complications. The common treatments for patients with symptomatic spinal metastases from RCC were surgery or radiation alone, surgery plus radiation, and a combination of surgery, radiation, and systemic therapy including chemotherapy, immunotherapy, and targeted therapy. Effect of targeted agents on bone metastases from RCC is currently limited.65 However, some studies reported that concomitant use of bisphosphonates and targeted agents may improve treatment efficacy.66,67 Stereotactic radiosurgery can be a more successful therapeutic modality than conventional radiotherapy.68,69
Patients with limited metastatic disease can be considered for metastasectomy, although they represent a minority (2-3%) of cases.70 Metastatic RCC can be treated via radical nephrectomy and resection, even in cases with multiple lesions if the number of lesions is small.71 Metastasectomy has clinical benefits for various prognostic groups and is independently associated with prolonged survival.72 Spinal metastasis, however, is considered a negative indicator of overall survival owing to the difficulty of surgical resection.73 The 5-year survival rate of patients with bone metastasis in the spine is 9%, compared with 30% in the appendicular skeleton.74 Osteolytic spinal metastasis from RCC often causes paralysis and intractable pain, thus reducing performance status. These findings suggest that complete removal of solitary spinal metastases, if achievable, can maintain performance status and better improve survival than other types of metastasectomies. We examined the clinical outcomes of 36 consecutive patients with RCC who underwent nephrectomy and complete removal of solitary spinal lesions between 1995 and 2010 at our institution.75 For all patients, the estimated median Cancer-specific survival (CSS) time was 130 months. The 3, 5, and 10-year CSS rates were 77.8%, 69.1%, and 58.0%, respectively, for all patients, and 72.7%, 54.5%, and 27.3%, respectively, for patients with lung metastases at the time of surgery. Only the presence of liver metastases was significantly associated with short-term survival after spinal metastasectomy. These results were more favorable than those previously reported.72,76,77 For selected patients, complete resection of solitary spinal metastases can potentially prolong survival.
THYROID CANCER‡
Thyroid carcinoma (TC) is generally not aggressive and is associated with a relatively favorable long-term survival.78 Bone metastases from differentiated TCs appear in 2–13% of patients.78,79 Spinal metastases are the most common type of thyroid bone metastases, accounting for approximately 50% of all bone metastases.80,81 Although thyroid spinal metastases from TC have the most favorable prognosis of all tumors metastasizing to the spine,82,83 they often cause intractable pain, and neurological deficits, thus substantially reducing the quality of life, and increase mortality. Moreover, most bone metastases from TC with osteolytic lesions are destructive and more resistant to systemic therapy and radiation than other organ metastases. Spinal metastases in particular result in pathologic fractures and spinal cord compression that substantially impair the performance status of patients.
Generally, radioiodine is the initial treatment for patients with TC metastases. However, radioiodine therapy may not be effective in bone metastases, especially with large lesions.84 Bisphosphonates or denosumab is the current standard of care for preventing skeletal complications associated with bone metastases. Since patients with spinal metastases from TC may have a long survival from their disease, it is essential to consider the potential advantages and disadvantages (osteonecrosis of the jaw and atypical femoral shaft fractures) of treating these patients with many years of therapy with these drugs. Conventional radiotherapy and stereotactic radiosurgery are standard treatment options for patients with localized bone pain, although TC bone metastases are generally radio-resistant.
The role of surgery for spinal metastases from TC is under significant revision. Old surgical techniques of decompression with or without stabilization resulted in poor outcomes, leading many to believe that radiotherapy was the preferred option over surgery.85 A significant proportion of patients with thyroid SMs have a solitary spinal lesion without non-spinal BMs nor other organ metastases and are eligible for aggressive surgical treatment, including metastasectomy, which is intended to improve their quality of life and prolong survival.86 The American Thyroid Association guidelines state that complete removal of BMs can prolong survival and is particularly appropriate for younger patients.87 Several studies reported the favorable postoperative outcomes of patients who underwent surgical removal of spinal metastases such as total en bloc spondylectomy.88,89 We examined the postoperative courses of 32 patients who underwent tumor excision surgery (complete excision group, 20; incomplete excision group, 12) for spinal metastases from TC with a minimum 4-year follow-up.90 Our results showed that patients in the complete excision group survived significantly longer than those in the incomplete excision group (5-year survival: 84% versus 50%; 10-year survival: 52% versus 8%), and all long-term survivors for > 18 months in the incomplete group experienced tumor recurrence and a consequent deterioration in performance status. Hence, complete surgical resection of spinal metastases, if achievable, has the potential not only to maintain performance status, but also to prolong survival.
OTHER TYPES OF CANCER‡
From other types of solid cancers including liver, gastric, colorectal, and bladder cancer, spinal metastases are rare.91 Especially liver and gastric cancers rarely present skeletal metastases with the incidence of approximately 5%.92,93 Most patients with spinal metastases from these cancers had advanced disease at the time of diagnosis, and isolated spinal metastases without the involvement of other organs such as the lung or liver was truly rare. Bone lesions are typically osteolytic. Many kinds of skeletal-related events (SREs) such as severe pain, pathologic fracture, spinal cord compression, and hypercalcemia may occur in cases of spinal metastases.94 Generally patients with spinal metastases from these cancers show a poor prognosis. Many patients developed SREs, and prevention and supportive management should be actively established to improve the quality of life in these patients.
Drug-based therapy using bisphosphonates or denosmab should be considered after diagnosis of spinal metastases to prevent SREs because the potential disadvantages of these drugs may be limited due to their short life-span estimated with disease condition.95,96 Patients with symptomatic spinal metastases can be treated with surgery, radiation, and a combination of these two options. The main goals of treatment are palliation (mainly for intractable pain), relief of neural compression (spinal cord and/or nerve roots) and local disease control. Conventional radiotherapy is standard treatment options for patients with localized bone pain. Oligometastases may be treated by stereotactic radiosurgery, and it showed better local control than conventional radiotherapy.97 Patients with life expectancies <3 months are generally regarded as unsuitable for surgery as the risks and drawbacks, including perioperative complications. Surgical interventions may be called for to reduce the risk of impending fracture, to treat pathological fracture, to reduce spinal instability and for the emergency treatment of spinal cord compression, with the primary goals of surgery being to preserve or restore skeletal integrity and function and to eliminate or prevent neurological compromise. For some patients without neurological deficits, less invasive surgical interventions such as kyphoplasty, vertebroplasty are advocated.
SUGGESTED READINGS
- Gage I, Recht A, Gelman R, et al. Long-term outcome following breast-conserving surgery and radiation therapy. Int J Radiat Oncol Biol Phys. 1995;33(2):245-251.
- Gasbarrini A, Cappuccio M, Mirabile L, et al. Spinal metastases: treatment evaluation algorithm. Eur Rev Med Pharmacol Sci. 2004;8(6):265-274.
- Santiago RJ, Wu L, Harris E, et al. Fifteen-year results of breast-conserving surgery and definitive irradiation for Stage I and II breast carcinoma: the University of Pennsylvania experience. Int J Radiat Oncol Biol Phys. 2004;58(1):233-240.
REFERENCES
- Harrington KD. Metastatic disease of the spine. J Bone Joint Surg Am. 1986;68(7):1110-1115.
- Tokuhashi Y, Matsuzaki H, Toriyama S, Kawano H, Ohsaka S. Scoring system for the preoperative evaluation of metastatic spine tumor prognosis. Spine. 1990;15(11):1110-1113.
- Tokuhashi Y, Matsuzaki H, Oda H, Oshima M, Ryu J. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine. 2005;30(19):2186-2191.
- Tokuhashi Y, Ajiro Y, Umezawa N. Outcome of treatment for spinal metastases using scoring system for preoperative evaluation of prognosis. Spine. 2009;34(1):69-73.
- Tomita K, Kawahara N, Kobayashi T, Yoshida A, Murakami H, Akamaru T. Surgical strategy for spinal metastases. Spine. 2001;26(3):298-306.
- Tomita K, Kawahara N, Murakami H, Demura S. Total en bloc spondylectomy for spinal tumors: improvement of the technique and its associated basic background. J Orthop Sci. 2006;11(1):3-12.
- Cappuccio M, Gasbarrini A, Van Urk P, Bandiera S, Boriani S. Spinal metastasis: a retrospective study validating the treatment algorithm. Eur Rev Med Pharmacol Sci. 2008;12(3):155-160.
- Fisher CG, DiPaola CP, Ryken TC, et al. A novel classification system for spinal instability in neoplastic disease: an evidence-based approach and expert consensus from the Spine Oncology Study Group. Spine. 2010;35(22):E1221-1229.
- Fourney DR, Frangou EM, Ryken TC, et al. Spinal instability neoplastic score: an analysis of reliability and validity from the spine oncology study group. J Clin Oncol. 2011;29(22):3072-3077.
- Boriani S, Weinstein JN, Biagini R. Primary bone tumors of the spine. Terminology and surgical staging. Spine. 1997;22(9):1036-1044.
- Kwan MK, Lee CK, Chan CY. Minimally invasive spinal stabilization using fluoroscopic-guided percutaneous screws as a form of palliative surgery in patients with spinal metastasis. Asian Spine J. 2016;10(1):99-110.
- Fourney DR, Schomer DF, Nader R, et al. Percutaneous vertebroplasty and kyphoplasty for painful vertebral body fractures in cancer patients. J Neurosurg. 2003;98(1 Suppl):21-30.
- Rajah G, Altshuler D, Sadiq O, Nyame VK, Eltahawy H, Szerlip N. Predictors of delayed failure of structural kyphoplasty for pathological compression fractures in cancer patients. J Neurosurg Spine. 2015;23(2):228-232.
- Iliopoulos P, Korovessis P, Vitsas V. PMMA embolization to the left dorsal foot artery during percutaneous vertebroplasty for spinal metastases. Eur Spine J. 2014;23 Suppl 2:187-191.
- Greenberg HS, Kim JH, Posner JB. Epidural spinal cord compression from metastatic tumor: results with a new treatment protocol. Ann Neurol. 1980;8(4): 361-366.
- Demura S, Kawahara N, Murakami H, et al. Surgical site infection in spinal metastasis: risk factors and countermeasures. Spine. 2009;34(6):635-639.
- Yokogawa N, Murakami H, Demura S, et al. Postoperative cerebrospinal fluid leakage associated with total en bloc spondylectomy. Orthopedics. 2015;38(7):e561-566.
- Seichi A, Kondoh T, Hozumi T, Karasawa K. Intraoperative radiation therapy for metastatic spinal tumors. Spine. 1999;24(5):470-475.
- Ryu S, Fang Yin F, Rock J, et al. Image-guided and intensity-modulated radiosurgery for patients with spinal metastasis. Cancer. 2003;97(8):2013-2018.
- Gerszten PC, Burton SA, Ozhasoglu C, Welch WC. Radiosurgery for spinal metastases: clinical experience in 500 cases from a single institution. Spine. 2007;32(2):193-199.
- Rosen LS, Gordon D, Tchekmedyian S, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: a phase III, double-blind, randomized trial—the Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21(16):3150-3157.
- Saad F, Gleason DM, Murray R, et al; Zoledronic Acid Prostate Cancer Study Group. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94(19);1458-1468.
- Stopeck AT, Lipton A, Body JJ, et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: a randomized, double-blind study. J Clin Oncol. 2010;28(35):5132-5139.
- Lu X, Kang Y. Organotropism of breast cancer metastasis. J Mammary Gland Biol Neoplasia. 2007;12(2-3):153-162.
- Berman AT, Thukral AD, Hwang WT, Solin LJ, Vapiwala N. Incidence and patterns of distant metastases for patients with early-stage breast cancer after breast conservation treatment. Clin Breast Cancer. 2013;13(2):88-94.
- Fowble BL, Solin LJ, Schultz DJ, Goodman RL. Ten year results of conservative surgery and irradiation for stage I and II breast cancer. Int J Radiat Oncol Biol Phys. 1991;21(2):269-277.
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 2005;365(9472):1687-1717.
- Briasoulis E, Karavasilis V, Kostadima L, Ignatiadis M, Fountzilas G, Pavlidis N. Metastatic breast carcinoma confined to bone: portrait of a clinical entity. Cancer. 2004;101(7):1524-1528.
- Shehadi JA, Sciubba DM, Suk I, et al. Surgical treatment strategies and outcome in patients with breast cancer metastatic to the spine: a review of 87 patients. Eur Spine J. 2007;16(8):1179-1192.
- Harvey HA. Issues concerning the role of chemotherapy and hormonal therapy of bone metastases from breast carcinoma. Cancer. 1997;80(8 Suppl):1646-1651.
- Saarto T, Vehmanen L, Blomqvist C, Elomaa I. Ten-year follow-up of 3 years of oral adjuvant clodronate therapy shows significant prevention of osteoporosis in early-stage breast cancer. J Clin Oncol. 2008;26(26):4289-4295.
- Chaichana KL, Pendleton C, Sciubba DM, Wolinsky JP, Gokaslan ZL. Outcome following decompressive surgery for different histological types of metastatic tumors causing epidural spinal cord compression. J Neurosurg Spine. 2009;11(1):56-63.
- Harris J, Morrow M, Norton L. Malignant tumors of the breast. In: DeVita VT, Hellman S, Rosenberg SA,eds. Cancer: Principles and Practice of Oncology. Philadelphia, PA: Lippincott-Raven; 1997:1557-1616.
- Hance KW, Anderson WF, Devesa SS, Young HA, Levine PH. Trends in inflammatory breast carcinoma incidence and survival: the surveillance, epidemiology, and end results program at the National Cancer Institute. J Natl Cancer Inst. 2005;97(13):966-975.
- Anderson WF, Chu KC, Chang S. Inflammatory breast carcinoma and noninflammatory locally advanced breast carcinoma: distinct clinicopathologic entities? J Clin Oncol. 2003;21(12):2254-2259.
- itera G, Probyn L, Ford M, et al. Correlation of computed tomography imaging features with pain response in patients with spine metastases after radiation therapy. Int J Radiat Oncol Biol Phys. 2011;81(3):827-830.
- Wu JS, Monk G, Clark T, Robinson J, Eigl BJ, Hagen N. Palliative radiotherapy improves pain and reduces functional interference in patients with painful bone metastases: a quality assurance study. Clin Oncol (R Coll Radiol). 2006;18(7):539-544.
- Diel IJ, Solomayer EF, Costa SD, et al. Reduction in new metastases in breast cancer with adjuvant clodronate treatment. N Engl J Med. 1998;339(6):357-363.
- Tancioni F, Navarria P, Mancosu P, et al. Surgery followed by radiotherapy for the treatment of metastatic epidural spinal cord compression from breast cancer. Spine. 2011;36(20):E1352-E1359.
- Roghmann F, Antczak C, McKay RR, et al. The burden of skeletal-related events in patients with prostate cancer and bone metastasis. Urol Oncol. 2015;33(1):17.e9-17.e18.
- DePuy V, Anstrom KJ, Castel LD, Schulman KA, Weinfurt KP, Saad F. Effects of skeletal morbidities on longitudinal patient-reported outcomes and survival in patients with metastatic prostate cancer. Support Care Cancer. 2007;15(7):869-876.
- Aass N, Fossa SD. Pre- and post-treatment daily life function in patients with hormone resistant prostate carcinoma treated with radiotherapy for spinal cord compression. Radiother Oncol. 2005;74(3):259-265.
- Rades D, Stalpers LJ, Veninga T, Rudat V, Schulte R, Hoskin PJ. Evaluation of functional outcome and local control after radiotherapy for metastatic spinal cord compression in patients with prostate cancer. J Urol. 2006;175(2):552-556.
- Bahl A, Hoefeler H, Duran I, et al. Health resource utilization associated with skeletal-related events in patients with advanced prostate cancer: a European subgroup analysis from an observational, multinational study. J Clin Med. 2014;3(3):883-896.
- Virtanen SS, Väänänen HK, Härkönen PL, Lakkakorpi PT. Alendronate inhibits invasion of PC-3 prostate cancer cells by affecting the mevalonate pathway. Cancer Res. 2002;62(9):2708-2714.
- Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastasis-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomised, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
- Vignani F, Bertaglia V, Buttigliero C, Tucci M, Scagliotti GV, Di Maio M. Skeletal metastases and impact of anticancer and bone-targeted agents in patients with castration-resistant prostate cancer. Cancer Treat Rev. 2016;44:61-73.
- Chen TC. Prostate cancer and spinal cord compression. Oncology. 2001;15(7):841-855.
- Crnalic S, Löfvenberg R, Bergh A, Widmark A, Hildingsson C. Predicting survival for surgery of metastatic spinal cord compression in prostate cancer: a new score. Spine. 2012;37(26):2168-2176.
- Shoskes DA , Perrin RG . The role of surgical management for symptomatic spinal cord compression in patients with metastatic prostate cancer. J Urol. 1989;142(2 Pt 1):337-339.
- Williams BJ, Fox BD, Sciubba DM, et al. Surgical management of prostate cancer metastatic to the spine. J Neurosurg Spine. 2009;10(5):414-422.
- Crnalic S, Hildingsson C, Wikström P, Bergh A, Löfvenberg R, Widmark A. Outcome after surgery for metastatic spinal cord compression in 54 patients with prostate cancer. Acta Orthop. 2012;83(1):80-86.
- Tsuya A, Kurata T, Tamura K, Fukuoka M. Skeletal metastases in non-small cell lung cancer: a retrospective study. Lung Cancer. 2007;57(2):229-232.
- Murakami H, Kawahara N, Demura S, Kato S, Yoshioka K, Tomita K. Total en bloc spondylectomy for lung cancer metastasis to the spine. J Neurosurg Spine. 2010;13(4):414-417.
- Morgen SS, Lund-Andersen C, Larsen CF, Engelholm SA, Dahl B. Prognosis in patients with symptomatic metastatic spinal cord compression: survival in different cancer diagnosis in a cohort of 2321 patients. Spine. 2013;38(16):1362-1367.
- Goodwin CR, Khattab MH, Sankey EW, et al. Factors associated with life expectancy in patients with metastatic spine disease from adenocarcinoma of the lung. Global Spine J. 2015;5(5):417-424.
- Lei M, Liu Y, Liu S, Wang L, Zhou S, Zhou J. Individual strategy for lung cancer patients with metastatic spinal cord compression. Eur J Surg Oncol. 2016;42(5):728-734.
- Park SJ, Lee CS, Chung SS. Surgical results of metastatic spinal cord compression (MSCC) from non-small cell lung cancer (NSCLC): analysis of functional outcome, survival time, and complication. Spine J. 2016;16(3):322-328.
- da Silva GT, Bergmann A, Santos Thuler LC. Prognostic factors in patients with metastatic spinal cord compression secondary to lung cancer: a systematic review of the literature. Eur Spine J. 2015;24(10):2107-2113.
- Tang Y, Qu J, Wu J, Li S, Zhou Y, Xiao J. Metastatic spinal cord compression from non-small-cell lung cancer treated with surgery and adjuvant therapies: a retrospective analysis of outcomes and prognostic factors in 116 patients. J Bone Joint Surg Am. 5;97(17):1418-1425.
- Zhao T, Gao Z, Wu W, He W, Yang YI. Effect of synchronous solitary bone metastasectomy and lung cancer resection on non-small cell lung cancer patients. Oncol Lett. 2016;11(3):2266-2270.
- Adiga GU, Dutcher JP, Larkin M, Garl S, Koo J. Characterization of bone metastases in patients with renal cell cancer. BJU Int. 2004;93(9):1237-1240.
- Lin PP, Mirza AN, Lewis VO, et al. Patient survival after surgery for osseous metastases from renal cell carcinoma. J Bone Joint Surg Am. 2007;89(8):1794-1801.
- Woodward E, Jagdev S, McParland L, et al. Skeletal complications and survival in renal cell cancer patients with bone metastases. Bone. 2011;48(1):160-166.
- Wood SL, Brown JE. Skeletal metastasis in renal cell carcinoma: current and future management options. Cancer Treat Rev. 2012;38(4):284-291.
- Keizman D, Ish-Shalom M, Pili R, et al. Bisphosphonates combined with sunitinib may improve the response rate, progression free survival and overall survival of patients with bone metastases from renal cell carcinoma. Eur J Cancer. 2012;48(7):1031-1037.
- Beuselinck B, Wolter P, Karadimou A, et al. Concomitant oral tyrosine kinase inhibitors and bisphosphonates in advanced renal cell carcinoma with bone metastases. Br J Cancer. 2012;107(10):1665-1671.
- Gerszten PC, Burton SA, Ozhasoglu C, et al. Stereotactic radiosurgery for spinal metastases from renal cell carcinoma. J Neurosurg Spine. 2005;3(4):288-295.
- Ghia AJ, Chang EL, Bishop AJ, et al. Single-fraction versus multifraction spinal stereotactic radiosurgery for spinal metastases from renal cell carcinoma: secondary analysis of Phase I/II trials. J Neurosurg Spine. 2016;24(5):829-836.
- Rini BI, Campbell SC, Escudier B. Renal cell carcinoma. Lancet. 2009;373(9669):1119-1132.
- Leibovich BC, Cheville JC, Lohse CM, et al.: A scoring algorithm to predict survival for patients with metastatic clear cell renal cell carcinoma: a stratification tool for prospective clinical trials. J Urol. 2005;174(5):1759-1763.
- Eggener SE, Yossepowitch O, Kundu S, Motzer RJ, Russo P. Risk score and metastasectomy independently impact prognosis of patients with recurrent renal cell carcinoma. J Urol. 2008;180(3):873-878.
- Kume H, Kakutani S, Yamada Y, et al. Prognostic factors for renal cell carcinoma with bone metastasis: who are the long-term survivors? J Urol. 2011;185(5):1611-1614.
- Jung ST, Ghert MA, Harrelson JM, Scully SP: Treatment of osseous metastases in patients with renal cell carcinoma. Clin Orthop Relat Res. 2003;(409):223-231.
- Kato S, Murakami H, Demura S, et al. Spinal metastasectomy of renal cell carcinoma: a 16-year single center experience with a minimum 3-year follow-up. J Surg Oncol. 2016;113(5):587-592.
- Donskov F, von der Maase H. Impact of immune parameters on long-term survival in metastatic renal cell carcinoma. J Clin Oncol. 2006;24(13):1997-2005.
- Naito S, Yamamoto N, Takayama T, et al. Prognosis of Japanese metastatic renal cell carcinoma patients in the cytokine era: a cooperative group report of 1463 patients. Eur Urol. 2010;57(2):317-325.
- Zettinig G, Fueger BJ, Passler C, et al. Long-term follow-up of patients with bone metastases from differentiated thyroid carcinoma - surgery or conventional therapy? Clin Endocrinol (Oxf). 2002;56(3):377-382.
- Muresan MM, Olivier P, Leclère J, et al. Bone metastases from differentiated thyroid carcinoma. Endocr Relat Cancer. 2008;15(1):37-49.
- Pittas AG, Adler M, Fazzari M, et al. Bone metastases from thyroid carcinoma: clinical characteristics and prognostic variables in one hundred forty-six patients. Thyroid. 2000;10(3):261-268.
- Farooki A, Leung V, Tala H, Tuttle RM. Skeletal-related events due to bone metastases from differentiated thyroid cancer. J Clin Endocrinol Metab. 2012;97(7):2433-2439.
- Quan GM, Pointillart V, Palussière J, Bonichon F. Multidisciplinary treatment and survival of patients with vertebral metastases from thyroid carcinoma. Thyroid. 2012;22(2):125-130.
- Hirabayashi H, Ebara S, Kinoshita T, et al. Clinical outcome and survival after palliative surgery for spinal metastases: palliative surgery in spinal metastases. Cancer. 2003;97(2):476-484.
- Hindié E, Zanotti-Fregonara P, Keller I, et al. Bone metastases of differentiated thyroid cancer: impact of early 131I-based detection on outcome. Endocr Relat Cancer. 2007;14(3):799-807.
- Choi D, Crockard A, Burger C, et al.; Global Spine Tumor Study Group. Review of metastatic spine tumour classification and indications for surgery: the consensus statement of the Global Spine Tumour Study Group. Eur Spine J. 2010;19(2):215-222.
- Kushchayeva YS, Kushchayev SV, Carroll NM, et al. Spinal metastases due to thyroid carcinoma: an analysis of 202 patients. Thyroid. 2014;24(10):1488-1500.
- Cooper DS, Doherty GM, Haugen BR, et al.; American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer. Revised American Thyroid Association management guideline for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19(11):1167-1214.
- Demura S, Kawahara N, Murakami H, et al. Total en bloc spondylectomy for spinal metastases in thyroid carcinoma. J Neurosurg Spine. 2011;14(2):172-176.
- Matsumoto M, Tsuji T, Iwanami A, et al. Total en bloc spondylectomy for spinal metastasis of differentiated thyroid cancers: a long-term follow-up. J Spinal Disord Tech. 2013;26(4):E137-E142.
- Kato S, Murakami H, Demura S, et al. The impact of complete surgical resection of spinal metastases on the survival of patients with thyroid cancer. Cancer Med. 2016;5(9):2343-2349. Epub 2016 Jul 19.
- Verlaan JJ, Choi D, Versteeg A, et al. Characteristics of patients who survived < 3 months or > 2 years after surgery for spinal metastases: can we avoid inappropriate patient selection? J Clin Oncol. 2016;34(25):3054-3061. Epub 2016 Jul 11.
- Goodwin CR, Yanamadala V, Ruiz-Valls A, et al. A systematic review of metastatic hepatocellular carcinoma to the spine. World Neurosurg. 2016;91:510-517.e4.
- Turkoz FP, Solak M, Kilickap S, et al. Bone metastasis from gastric cancer: the incidence, clinicopathological features, and influence on survival. J Gastric Cancer. 2014;14(3):164-172.
- Coleman RE. Skeletal complication of malignancy. Cancer. 1997;80(8 Suppl):1588-1594.
- Aapro M, Abrahamsson PA, Body JJ, et al. Guidance on the use of bisphosphonates in solid tumours: recommendations of an international expert panel. Ann Oncol. 2008;19(3):420-432.
- Santini D, Tampellini M, Vincenzi B, et al. Natural history of bone metastases in colorectal cancer: final results of a large Italian bone metastases study. Ann Oncol. 2012;23(8):2072-2077.
- Chang UK, Kim MS, Han CJ, Lee DH. Clinical result of stereotactic radiosurgery for spinal metastasis from hepatocellular carcinoma: comparison with conventional radiation therapy. J Neurooncol. 2014;119(1):141-148.